 |
 |
- Search
AbstractObjectivesAmong the inflammatory mediators, phorbol 12-myristate 13-acetate (PMA) is associated with the regulation of MUC5B expression in the airway epithelial cells. Epigallocatechin-3-gallate (EGCG) is the major component of green tea extract. The biological activity of EGCG includes reduction of cholesterol and antioxidant activity, as well as anti-inflammatory effect. However, the precise action mechanism of anti-inflammatory effect of EGCG in the airway epithelial cells has not been fully defined. This study investigates the effect and the brief signaling pathway of EGCG on PMA-induced MUC5B expression in the airway epithelial cells.
MethodsIn NCI-H292 airway epithelial cells, the effect and signaling pathway of EGCG on MUC5B expression were investigated using real-time polymerase chain reaction analysis, enzyme immunoassay, immunohistochemical analysis, gelatin zymography assay, and immunoblot analysis.
ResultsIn NCI-H292 airway epithelial cells, PMA induced MUC5B expression, phosphorylation of p38 mitogen-activated protein kinase (MAPK), and matrix metalloproteinase-9 (MMP-9) expression and protein activity. EGCG significantly decreased PMA-induced MUC5B expression, phosphorylation of p38 MAPK, and MMP-9 expression and protein activity. SB203580 (p38 MAPK inhibitor) significantly decreased PMA-induced MMP-9 expression. In addition, SB203580 and MMP-9 I (MMP-9 inhibitor) significantly decreased PMA-induced MUC5B expression.
Mucus plays an important role in protecting the human airway from external environment. Mucins are highly glycosylated proteins that are the major components of mucus, and which are responsible for its viscoelastic properties [1]. Mucins are subdivided into secretory and membrane associated forms [2]. Among the mucins, MUC5AC, MUC5B, and MUC8 are representative secretory mucin genes in the human airway [3]. In the airway inflammatory diseases, such as asthma and bronchitis, airway mucins are produced and secreted in increased quantities. Furthermore, excessive mucus may become more densely packed and can lead to airway closure [4].
Phorbol 12-myristate 13-acetate (PMA), a protein kinase C activator, has been used as an inflammatory stimulant to express mucin genes in airway epithelial cells: PMA has been reported to up-regulate MUC5B expression via matrix metalloproteinase-9 (MMP-9) in airway epithelial cells [5,6]. Epigallocatechin-3-gallate (EGCG) is the major component of green tea extract. The various functions of EGCG include reduction of cholesterol and antioxidant activity, which prevents cellular injury due to oxidative stress [7-9]. Recently, several studies reported the anti-allergic or anti-inflammatory effect of EGCG: EGCG can counteract allergic asthma-like reaction by modulating nitric oxide synthase activity [10]. EGCG regulates inflammatory cell migration by suppressing MMP-9 production and reactive oxygen species generation [11]. However, the precise mechanism of action about anti-inflammatory effect of EGCG has not been fully defined.
Therefore, this study aimed to investigate the effect and the brief signaling pathway of EGCG on PMA-induced MUC5B expression in human airway epithelial cells.
EGCG and PMA were obtained from Sigma-Aldrich (St Louis, MO, USA). Mucin-producing human NCI-H292 airway epithelial cells were obtained from the American Type Culture Collection (Manassas, VA, USA). RPMI 1640 medium and Trizol were obtained from Invitrogen (Carlsbad, CA, USA). Real-time polymerase chain reaction (PCR) kits were obtained from Roche Applied Science (Mannheim, Germany). Fetal bovine serum (FBS) was obtained from Hyclone Laboratories (Logan, UT, USA). Primary antibody and anti-goat or anti-mouse horseradish peroxidase (HRP)-conjugated secondary antibody of MUC5B, monoclonal human MMP-9 antibody, and ╬▓-actin were obtained from Santa Cruz Biotechnology (Santa Cruz, CA, USA). Extracellular signal related kinase 1/2 (ERK1/2), phospho-ERK1/2, p38, and phospho-p38 antibodies were purchased from Cell Signaling Technology (Danvers, MA, USA). SB203580, a specific inhibitor for p38 mitogen-activated protein kinase (MAPK), was purchased from Biomol (Plymouth Meeting, PA, USA). MMP-9 I (MMP-9 inhibitor), a specific inhibitor for MMP-9, was purchased from Calbiochem (Frankfurt, Germany).
NCI-H292 airway epithelial cells were cultured in RPMI 1640 medium supplemented with 2 mM L-glutamine, 100 U/mL penicillin, 100 ┬Ąg/mL streptomycin, and 10% FBS. The cells were grown at 37Ōäā in 5% CO2 fully humidified air. When confluent, the cells were incubated in RPMI 1640 medium containing 0.5% FBS for 24 hours. The cells were then rinsed with serum-free RPMI 1640 medium and exposed to the indicated concentrations of EGCG for 1 hour after pretreatment with PMA. This study was approved by the institutional review board at the Yeungnam University Medical Center.
Real-time PCR was performed with the LC Fast Start DNA Master SYBR Green kit (Roche Applied Science) using 0.5 ┬ĄL of cDNA, corresponding to 25 ng of total RNA in a 10 ┬ĄL final volume, 2.5 mM MgCl2 and 0.5 ┬ĄM of each primer (final concentration). Quantitative PCR was performed using a LightCycler (Roche Applied Science) for 45 cycles at 95Ōäā for 10 seconds, specific annealing temperature for 5 seconds and 72Ōäā for 10 seconds. Data were normalized to glyceraldehydes-3-phosphate dehydrogenase (GAPDH). Amplification specificity was evaluated using melting curve, following the manufacturer's instructions (Roche Applied Science). Primer sequences and conditions were used according to previously published studies of MUC5B, MMP-9, and GAPDH [6,12].
MUC5B protein level was determined by an enzyme-linked immunosorbent assay (ELISA). Samples of cell lysates from NCI-H292 airway epithelial cells were incubated at 40Ōäā in wells of 96-well plate. Plates were blocked with 2% bovine serum albumin and incubated with primary antibody of MUC5B diluted with PBS containing 0.05% Tween 20 for 1 hour. The wells were treated with HRP-conjugated secondary antibody of MUC5B into each well. After 4 hours, color was developed using 3, 3', 5, 5'-tetramethylbenzidine peroxidase solution and stopped with 2N-H2SO4. Optical density measurements were obtained from an ELISA reader (EL800; Bio-Tek Instruments, Winooski, VT, USA) at 450 nm.
MMP-9 protein activity was measured by gelatin zymography. In brief, the supernatant of cultured NCI-H292 airway epithelial cells containing 10 ┬Ąg of protein were mixed with 2├Śsodium-dodecyl sulfate (SDS) loading buffer before being loaded into 10% polyacrylamide gels (wt/vol) contain 0.1% (wt/vol) gelatin as a substrate (Biotech, Piscataway, NJ, USA). The protein was subjected to electrophoresis at 80 volt for 120 minutes and the gel was then washed with zymogram renaturing solution (Biotech). The gel was incubated in zymogram developing solution (Biotech) for 24 hours. After incubation, the gel was stained by 0.5% Coomassie Blue R-250 (Bio-Rad Lab., Hercules, CA, USA) and destained in 30% methanol and 5% acetic acid solution, and then rinsed to visualize digested bands in the gelatin matrix. The gels were photographed and the averages of band intensity were measured using commercially available imaging software (Scion Co., Frederick, MD, USA).
Samples of cell from NCI-H292 airway epithelial cells were seeded in wells of a 6-well plate and treated with EGCG. The cells were exposed to trypsin, and formed into pellets at 700 g at 4Ōäā and pellets were resuspended in lysis buffer. The preparation was then clarified by centrifugation, and the supernatant was saved as a whole-cell lysate. Proteins (20 ┬Ąg) were separated using 10% reducing SDS-polyacrylamide gel electrophoresis and electroblotted onto a nitrocellulose membrane. The membrane was blocked with 5% nonfat dry milk, followed by incubation with the indicated primary antibody of p38, ERK1/2, or human monoclonal MMP-9 antibody for 4 hours. Subsequently, the membrane was incubated for 1 hour with secondary antibody of p38, ERK1/2, or human monoclonal MMP-9 antibody conjugated to HRP, and developed using an enhanced chemiluminescence kit (Perkin Elmer Life Sciences). Bands were detected after exposure to X-ray film for 10 seconds.
To investigate the effect of EGCG on PMA-induced MUC5B expression, NCI-H292 airway epithelial cells were incubated with PMA (10 nM) in the absence or presence (10, 50, and 100 ┬ĄM) of EGCG. The results of real-time PCR and ELISA showed that PMA induced MUC5B mRNA expression and protein production, which were significantly decreased by pretreatment with EGCG (Fig. 1).
To investigate the MAPK signaling pathway of PMA-induced MUC5B expression, NCI-H292 airway epithelial cells were incubated with PMA (10 nM) and phosphorylation of ERK1/2 and p38 MAPK were analyzed by Western blot. As time elapsed, phosphorylation of p38 MAPK was significantly increased: phosphorylation of p38 MAPK peaked at 20 minutes after treatment with PMA, but phosphorylation of ERK1/2 did not increase (Fig. 2A). To investigate the effect of EGCG on PMA-induced phosphorylation of p38 MAPK, NCI-H292 airway epithelial cells were pretreated with EGCG (10, 50, and 100 ┬ĄM) for 1 hour before being exposed to PMA (10 nM). The result of Western blot showed that EGCG significantly decreased PMA-induced phosphorylation of p38 MAPK (Fig. 2B).
To investigate the signaling pathway associated with MMP-9, NCI-H292 airway epithelial cells were incubated with PMA (10 nM) in the absence or presence (50 and 100 ┬ĄM) of EGCG. The result of real-time PCR showed that PMA induced MMP-9 mRNA expression and EGCG significantly decreased PMA-induced MMP-9 mRNA expression (Fig. 3A). The result of gelatin zymography showed that EGCG inhibited PMA-induced MMP-9 protein activity (Fig. 3B). To investigate the correlation between MMP-9 and p38 MAPK, the cells were pretreated with SB203580 as a p38 MAPK inhibitor (10 and 20 ┬ĄM) for 1 hour before being exposed to PMA (10 nM). The result of real-time PCR showed that SB203580 significantly decreased PMA-induced MMP-9 mRNA expression (Fig. 3C).
To investigate the brief signaling pathway of EGCG on PMA-induced MUC5B expression, NCI-H292 airway epithelial cells were pretreated with SB203580 as a p38 MAPK inhibitor (20 ┬ĄM) or MMP-9 I as a MMP-9 I (5 nM) for 1 hour before being exposed to PMA (10 nM). The result of real-time PCR showed that SB203580 and MMP-9 I significantly decreased PMA-induced MUC5B mRNA expression (Fig. 4).
PMA, a protein kinase C activator, has been used as an inflammatory stimulant to modulate various cellular events. PMA also plays a significant role in the induction of MUC5AC and MUC5B in NCI-H292 airway epithelial cells [5,6]. PMA-induced MUC5B expression is regulated through epidermal growth factor receptor and p38-mediated transcriptional mechanism [6,13,14]. In this study, PMA induced MUC5B expression and phosphorylation of p38 MAPK in NCI-H292 airway epithelial cells, and EGCG significantly decreased PMA-induced phosphorylation of p38 MAPK. These results suggest that p38 MAPK plays a role in the signal pathway of PMA-induced MUC5B expression, and EGCG can down-regulate MUC5B expression in NCI-H292 airway epithelial cells.
MMP-9 is a kind of collagenase that cleaves type IV collagen, which makes up a substantial portion of the basement membrane of airways. MMP-9 plays a role in tissue remodeling in diseases such as asthma and bronchiectasis; increased MMP-9 activity in bronchoalveolar lavage fluid was observed in asthmatic patients compared to healthy subjects [15]. Several studies have demonstrated that MMP-9 mediates mucin gene expression in airway epithelial cells. Acrolein-activated MMP-9, leading to MUC5AC accumulation, contributes to persistent mucin production in chronic obstructive pulmonary disease [16]. Lipopolysaccharide-induced MUC5AC production may be partially mediated by MMP-9 activation [12,17]. Doxycycline inhibits PMA-induced MUC5B expression through the MMP-9 and p38 pathways in human airway epithelial cells [6]. In this study, PMA induced MMP-9 mRNA expression and SB203580 significantly decreased PMA-induced MMP-9 mRNA expression. These results suggest that MMP-9 may be an important mediator of PMA-induced MUC5B expression, and p38 MAPK plays a role in PMA-induced MMP-9 expression.
For the perspective of the protective effect of EGCG on allergic or inflammatory reaction in airway epithelial cells, EGCG can sustain endothelial nitric oxide synthase activity and maintain endogenous nitric oxide generation at physiological levels. This can counteract the activation of inducible nitric oxide synthase, which releases excess nitric oxide and subsequently activates cyclooxygenase-2 to produce inflammatory prostaglandins [10]. In allergic or inflammatory reaction in airway epithelial cells, EGCG reduced the pathologic lung damage due to the suppression of reactive oxygen species generation through the inhibition of inflammatory cells migration by reducing MMP-9 expression and also by suppressing tumor necrosis factor-╬▒ and interleukin-5 production [12]. And EGCG reduced inflammatory cell recruitment and overall inflammatory response in airways and lung tissues by directly binding to the proinflammatory chemokines, attenuating their chemotactic abilities [18]. Presently, the results of this study showed that EGCG significantly decreased PMA-induced MUC5B expression, phosphorylation of p38 MAPK, and MMP-9 expression and protein activity. SB203580 (p38 MAPK inhibitor) significantly decreased PMA-induced MMP-9 expression. In addition, SB203580 and MMP-9 I significantly decreased PMA-induced MUC5B expression. These suggest that EGCG can down-regulate PMA-induced MUC5B expression, which is associated with p38 MAPK dependent MMP-9 signaling pathway (Fig. 5).
In conclusion, the results of this study demonstrate that EGCG down-regulates PMA-induced MUC5B expression through the p38 MAPK dependent MMP-9 signaling pathway in human airway epithelial cells. Therefore, EGCG may have protective effect on the airway inflammatory diseases through the regulation of mucin gene expression in airway epithelial cells. And EGCG could be a novel therapeutic agent for controlling the airway inflammatory diseases.
References1. Williams OW, Sharafkhaneh A, Kim V, Dickey BF, Evans CM. Airway mucus: from production to secretion. Am J Respir Cell Mol Biol. 2006 5;34(5):527-536. PMID: 16415249.
2. Ali MS, Pearson JP. Upper airway mucin gene expression: a review. Laryngoscope. 2007 5;117(5):932-938. PMID: 17473699.
3. Evans CM, Kim K, Tuvim MJ, Dickey BF. Mucus hypersecretion in asthma: causes and effects. Curr Opin Pulm Med. 2009 1;15(1):4-11. PMID: 19077699.
4. Rose MC, Voynow JA. Respiratory tract mucin genes and mucin glycoproteins in health and disease. Physiol Rev. 2006 1;86(1):245-278. PMID: 16371599.
5. Yuan-Chen Wu D, Wu R, Reddy SP, Lee YC, Chang MM. Distinctive epidermal growth factor receptor/extracellular regulated kinase-independent and -dependent signaling pathways in the induction of airway mucin 5B and mucin 5AC expression by phorbol 12-myristate 13-acetate. Am J Pathol. 2007 1;170(1):20-32. PMID: 17200179.
6. Bae CH, Chen SM, Lee HM, Song SY, Kim YD. The effect of doxycycline on PMA-induced MUC5B expression via MMP-9 and p38 in NCI-H292 cells. Clin Exp Otorhinolaryngol. 2011 12;4(4):177-183. PMID: 22232712.
7. Raederstorff DG, Schlachter MF, Elste V, Weber P. Effect of EGCG on lipid absorption and plasma lipid levels in rats. J Nutr Biochem. 2003 6;14(6):326-332. PMID: 12873714.
8. Oliva J, Bardag-Gorce F, Tillman B, French SW. Protective effect of quercetin, EGCG, catechin and betaine against oxidative stress induced by ethanol in vitro. Exp Mol Pathol. 2011 6;90(3):295-299. PMID: 21352821.
9. Lee H, Bae JH, Lee SR. Protective effect of green tea polyphenol EGCG against neuronal damage and brain edema after unilateral cerebral ischemia in gerbils. J Neurosci Res. 2004 9;77(6):892-900. PMID: 15334607.
10. Bani D, Giannini L, Ciampa A, Masini E, Suzuki Y, Menegazzi M, et al. Epigallocatechin-3-gallate reduces allergen-induced asthma-like reaction in sensitized guinea pigs. J Pharmacol Exp Ther. 2006 6;317(3):1002-1011. PMID: 16525038.
11. Kim SH, Park HJ, Lee CM, Choi IW, Moon DO, Roh HJ, et al. Epigallocatechin-3-gallate protects toluene diisocyanate-induced airway inflammation in a murine model of asthma. FEBS Lett. 2006 3;580(7):1883-1890. PMID: 16516891.
12. Wang Y, Shen Y, Li K, Zhang P, Wang G, Gao L, et al. Role of matrix metalloproteinase-9 in lipopolysaccharide-induced mucin production in human airway epithelial cells. Arch Biochem Biophys. 2009 6;486(2):111-118. PMID: 19389382.
13. Hewson CA, Edbrooke MR, Johnston SL. PMA induces the MUC5AC respiratory mucin in human bronchial epithelial cells, via PKC, EGF/TGF-alpha, Ras/Raf, MEK, ERK and Sp1-dependent mechanisms. J Mol Biol. 2004 11;344(3):683-695. PMID: 15533438.
14. Wu DY, Wu R, Chen Y, Tarasova N, Chang MM. PMA stimulates MUC5B gene expression through an Sp1-based mechanism in airway epithelial cells. Am J Respir Cell Mol Biol. 2007 11;37(5):589-597. PMID: 17600309.
15. Ko FW, Diba C, Roth M, McKay K, Johnson PR, Salome C, et al. A comparison of airway and serum matrix metalloproteinase-9 activity among normal subjects, asthmatic patients, and patients with asthmatic mucus hypersecretion. Chest. 2005 6;127(6):1919-1927. PMID: 15947303.
16. Deshmukh HS, Shaver C, Case LM, Dietsch M, Wesselkamper SC, Hardie WD, et al. Acrolein-activated matrix metalloproteinase 9 contributes to persistent mucin production. Am J Respir Cell Mol Biol. 2008 4;38(4):446-454. PMID: 18006877.
17. Ren S, Guo LL, Yang J, Liu DS, Wang T, Chen L, et al. Doxycycline attenuates acrolein-induced mucin production, in part by inhibiting MMP-9. Eur J Pharmacol. 2011 1;650(1):418-423. PMID: 21036164.
18. Qin S, Alcorn JF, Craigo JK, Tjoeng C, Tarwater PM, Kolls JK, et al. Epigallocatechin-3-gallate reduces airway inflammation in mice through binding to proinflammatory xhemokines and inhibiting inflammatory cell recruitment. J Immunol. 2011 3;186(6):3693-3700. PMID: 21307292.
Fig.┬Ā1The effect of EGCG on PMA-induced MUC5B expression. To investigate the effect of EGCG on PMA-induced MUC5B expression, NCI-H292 airway epithelial cells were incubated with PMA in the absence or presence of EGCG. (A) Real-time PCR and (B) ELISA showed that PMA induced MUC5B mRNA expression and protein production, which were significantly decreased by pretreatment with EGCG. EGCG, epigallocatechin-3-gallate; ELISA, enzyme-linked immunosorbent assay; PCR, polymerase chain reaction; PMA, phorbol 12-myristate 13-acetate. *P<0.05 compared with zero value. **P<0.05 compared with PMA alone. 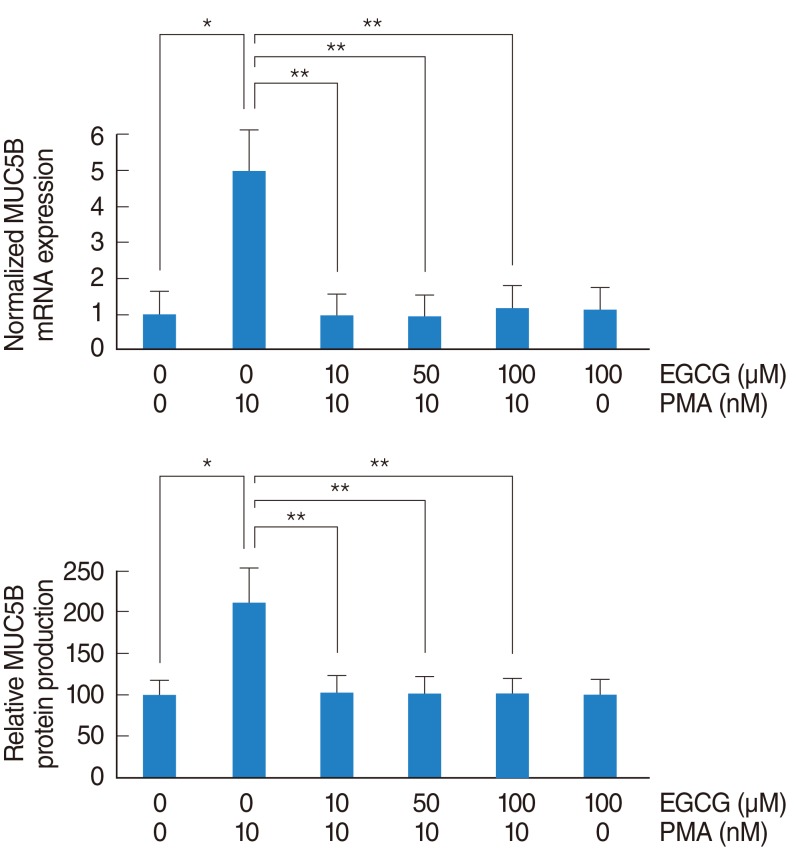
Fig.┬Ā2The effect of EGCG on PMA-induced phosphorylation of p38 MAPK. To investigate the MAPK signaling pathway of PMA-induced MUC5B expression, NCI-H292 airway epithelial cells were incubated with PMA in the absence or presence of EGCG and phosphorylation of ERK1/2 and p38 MAPK were analyzed by Western blot. (A) As time elapsed, phosphorylation of p38 MAPK was significantly increased: phosphorylation of p38 MAPK peaked at 20 minutes after treatment with PMA, but phosphorylation of ERK1/2 did not increase. (B) EGCG significantly decreased PMA-induced phosphorylation of p38 MAPK. EGCG, epigallocatechin-3-gallate; ERK1/2, extracellular signal related kinase 1/2; MAPK, mitogen-activated protein kinase; PMA, phorbol 12-myristate 13-acetate. *P<0.05 compared with zero value. **P<0.05 compared with PMA alone. 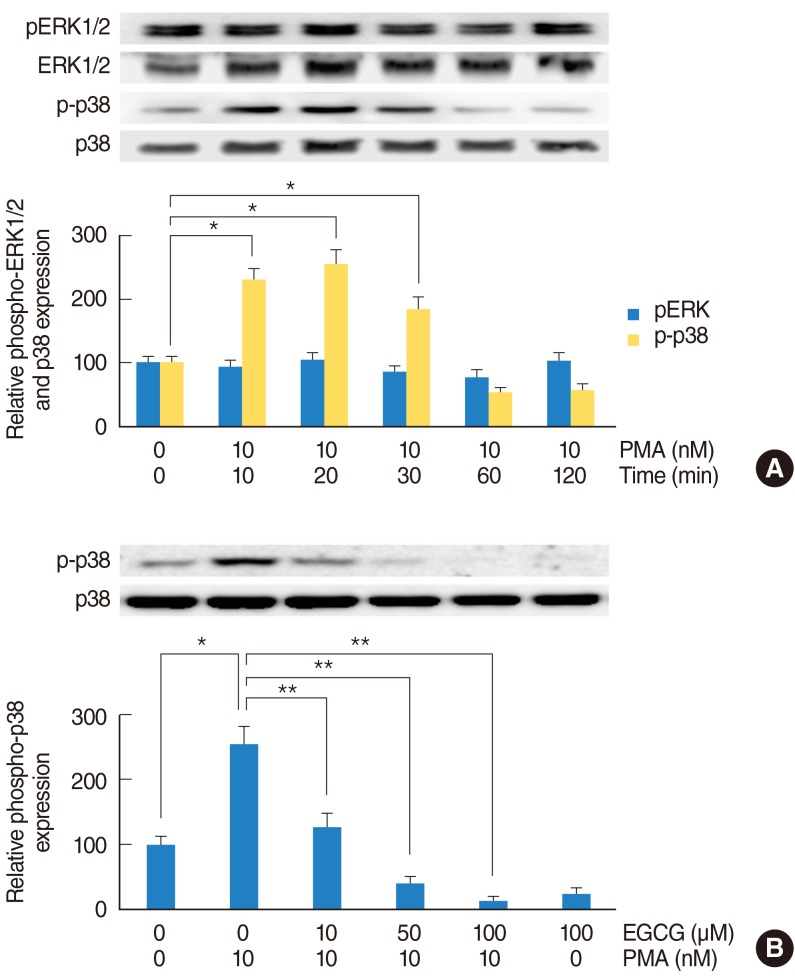
Fig.┬Ā3The effect of EGCG on PMA-induced MMP-9 expression. To investigate the signaling pathway associated with MMP-9, NCI-H292 airway epithelial cells were incubated with PMA in the absence or presence of EGCG. (A) Real-time PCR showed that PMA induced MMP-9 mRNA expression and EGCG significantly decreased PMA-induced MMP-9 mRNA expression. (B) Gelatin zymography showed that EGCG inhibited PMA-induced MMP-9 protein activity. To investigate the correlation between MMP-9 and p38 MAPK, NCI-H292 airway epithelial cells were pretreated with SB203580 before being exposed to PMA. (C) Real-time PCR showed that SB203580 significantly decreased PMA-induced MMP-9 mRNA expression. EGCG, epigallocatechin-3-gallate; MAPK, mitogen-activated protein kinase; MMP-9, matrix metalloproteinase-9; PCR, polymerase chain reaction; PMA, phorbol 12-myristate 13-acetate. *P<0.05 compared with zero value. **P<0.05 compared with PMA alone. 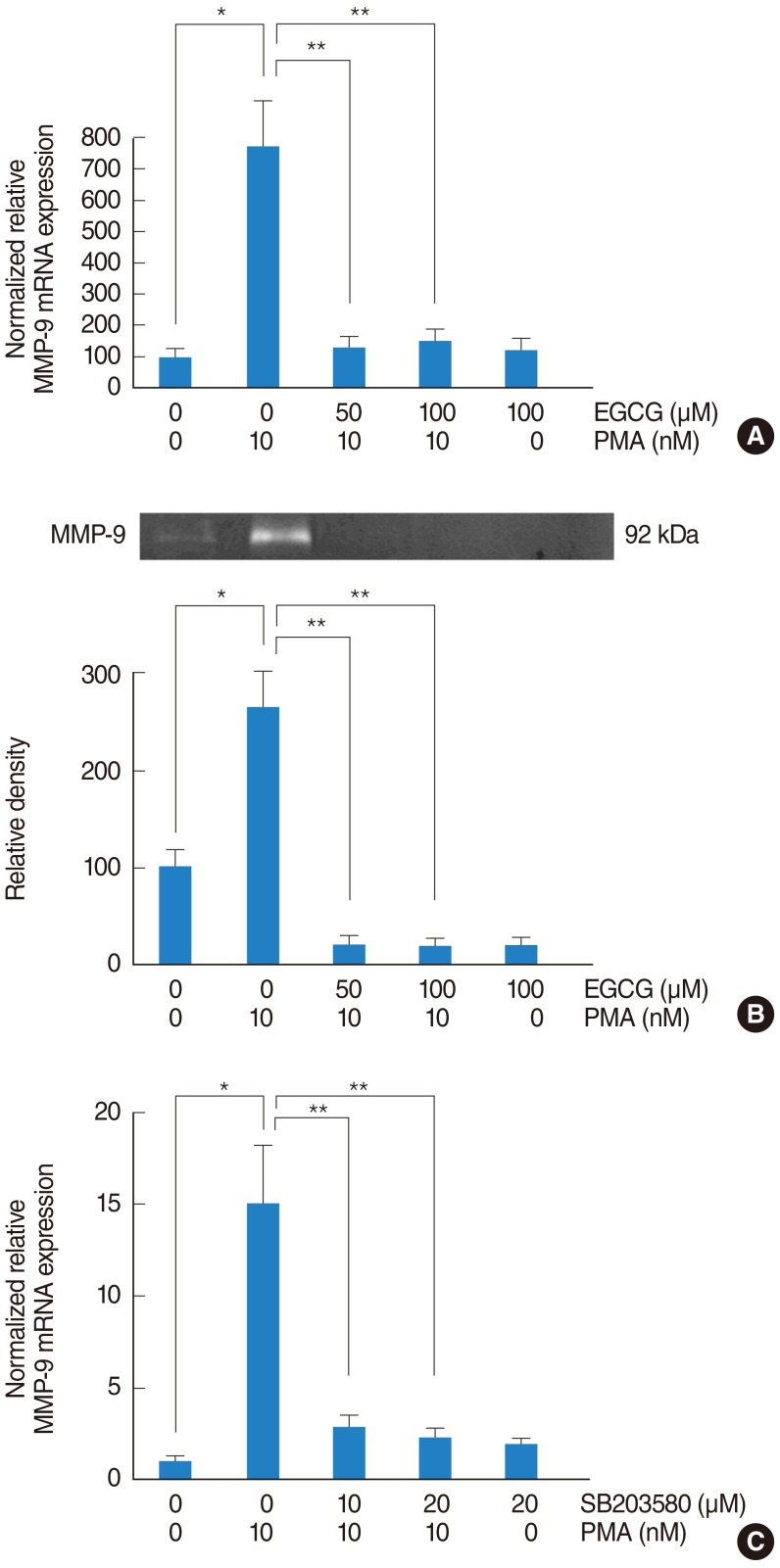
Fig.┬Ā4The signaling pathway of EGCG on PMA-induced MUC5B expression. To investigate the brief signaling pathway of EGCG on PMA-induced MUC5B expression, NCI-H292 airway epithelial cells were pretreated with SB203580 as a p38 MAPK inhibitor or MMP-9 I as a MMP-9 inhibitor before being exposed to PMA. Real-time PCR showed that SB203580 and MMP-9 I significantly decreased PMA-induced MUC5B mRNA expression. EGCG, epigallocatechin-3-gallate; MAPK, mitogen-activated protein kinase; MMP-9 I, matrix metalloproteinase-9 inhibitor; PCR, polymerase chain reaction; PMA, phorbol 12-myristate 13-acetate. *P<0.05 compared with zero value. **P<0.05 compared with PMA alone. 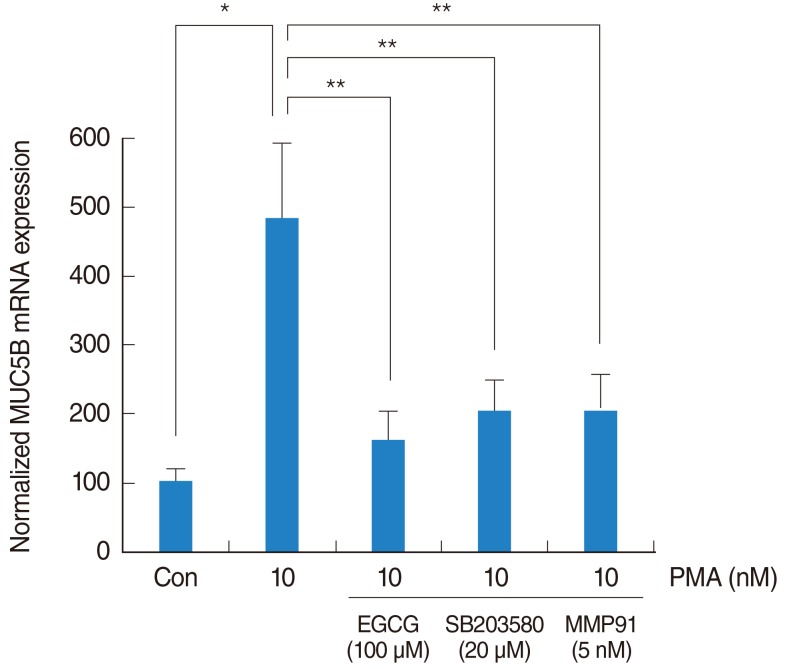
Fig.┬Ā5The schematic signaling pathway of EGCG on PMA-induced MUC5B expression. EGCG can down-regulate PMA-induced MUC5B expression, which is associated with p38 MAPK dependent MMP-9 signaling pathway. PMA, phorbol 12-myristate 13-acetate; EGCG, epigallocatechin-3-gallate; MAPK, mitogen-activated protein kinase; MMP-9, matrix metalloproteinase-9. 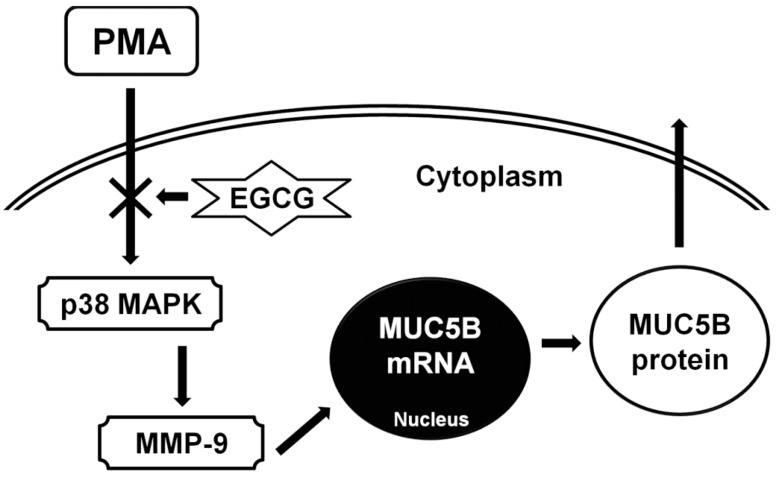
|
|
||||||||||||||||||||||||||||||||||||







