INTRODUCTION
Hearing-impaired children who receive intervention during the first 6 to 12 months of life have been shown to develop better speech and language skills than the children who receive delayed intervention (1, 2). Due to the importance of the early identification and management of congenital hearing loss, new-born hearing screening programs have been set up in many countries, and more hearing-impaired babies are now being identified during the neonatal term than ever before (3).
Evoked potential testing is critical for determining the audiologic thresholds in babies who are too young for traditional behavioral methods, including visual reinforcement audiometry and conditional play audiometry because the behavioral audiometry methods are neither accurate nor reliable before 6 months of age (4). The most commonly used click-auditory brainstem response (C-ABR) test paradigm applies acoustic click stimuli, and these may generate synchronous neural firing in the auditory pathway and this correlates with the best or average threshold in the 1-4 kHz range (5). Yet detailed information concerning the frequency-specific thresholds cannot be obtained, and the hearing loss that's restricted to particular frequencies may be overlooked (6). Moreover, the maximum presentation level of a click is limited, which makes it difficult to differentiate between severe and profound hearing loss. C-ABR recordings are limited by the subjective visual inspection method that's used to determine the threshold (7, 8).
The auditory steady state response (ASSR) is an alternative evoked potential technique that uses periodic electrical responses of the brain to auditory stimuli that are presented at a fast enough rate for eliciting successive responses (9, 10). These tones are reasonably frequency-specific because the continuous tonal stimuli contain energy in a much smaller frequency range than do clicks. The ASSR is elicited in response to sinusoidal amplitude and/or frequency-modulated tones (11). The best modulation rates for audiometric purposes seems to be between 75 and 110 Hz because at these rates the problems associated with sleeping can be avoided (12) and babies can be reliably recorded during sleep (13). Another particular advantage of ASSR is that the stimulus tone can be presented at high levels up to 120 dB HL at most frequencies and these levels can estimate the ears with minimal residual hearing (8). Furthermore, in contrast with the C-ABR, the steady-state responses can be measured objectively and automatically by a computer.
We have therefore investigated the degree to which the ASSR thresholds correlate with the C-ABR thresholds in sedated infants who are younger than 1 yr old and who have a range of hearing impairments, and we used protocols that may be feasible in routine clinical practice because the parameters used for data collection and analysis vary widely according to the individual clinic. We also tested the effectiveness of the ASSR as a measure of hearing sensitivity in infants who are suspected of having significant congenital hearing loss.
MATERIALS AND METHODS
Subjects
For this study, we included all the infants who were referred to the Department of Otolaryngology of Asan Medical Center for C-ABR testing under sedation with chloral hydrate from February 2004 through December 2006. They consisted of 76 infants (46 boys and 30 girls, a total 151 ears) who ranged in age from 1 to 12 months (average age: 5.7 months) at the time of C-ABR and ASSR testing. All the participants suspected of having sensorineural hearing loss had been referred from several local clinics after the infants failed newborn screening assessments with using automated electrophysiologic techniques. Before the auditory evaluations, middle ear inspection and tympanometry were done to rule out conductive hearing loss such as otitis media. For comparing the ASSR results between infants and adults with normal hearing, we analyzed 39 ears of infants (10 boys and 10 girls, mean age: 6.2 months) and 39 ears of an adult group (11 males and 9 females, mean age: 31.2 yr), and all the ears' hearing thresholds were less than 20 dB nHL by C-ABR and the otoscopic findings were normal.
The study protocol was approved by the Institutional Review Board of Asan Medical Center, and written informed consent was obtained from the parents of each subject.
Audiologic evaluation
C-ABR
The C-ABR was recorded using the Bio-logic Navigator Pro with TDH-39 headphones and ER-3 insert earphones with foam earplugs. Surface recording electrodes were applied to the high forehead (active), low forehead (ground) and the mastoids (reference) of both ears. The electrode impedance values were less than 5 kohms. The click rarefaction polarity stimuli consisted of 100 ┬Ąs pulses of a maximum of 90 dB nHL at a rate of 13.1/sec and a filter set of 100-3,000 Hz bandwidth with an amplifier gain of 10,000. Time windows of 15 msec were used to record the C-ABR. At each presentation level, a minimum of 1,024 sweeps was averaged. A 5 dB increment or decrement was used to determine the threshold. Threshold was defined as the lowest level at which a C-ABR was present, as determined by visual inspection of the waveforms displayed on the computer screen. The lowest C-ABR thresholds were assessed in 20 adult ears with normal hearing (the hearing thresholds of the behavioral test were less than 20 dB HL for 0.5, 1, 2, and 4 kHz) for the value of 0 dB nHL.
ASSR
The frequency-specific thresholds were recorded by a Bio-logic MASTER system with TDH-39 headphones and ER-3A insert earphones with foam earplugs. The stimuli used to elicit the ASSR consisted of carrier frequencies of 0.5, 1, 2, and 4 kHz, and these were 100% amplitude modulated and 20% frequency modulated at modulation frequencies of 82, 84, 87, 89, 91, 94, 96, and 99 Hz (Ōēż80 dB HL intensity level) and 67 and 69 Hz (Ōēź90 dB HL intensity level), respectively. The initial intensity of the ASSR was 60 dB HL. The maximum presentation levels were 115 dB HL for frequencies of 0.5, 1, 2, and 4 kHz. Between 16 and 32 sweeps were analyzed during each recording. The data was averaged in the time domain and then submitted to fast Fourier transform analysis. The significant response level was monitored after each sweep and it was set at P<0.05. Recording electrodes were placed at the forehead (active), shoulder (ground) and the nape of the neck (ground). The electrode impedance values were less than 10 kohms. The ASSR thresholds were determined at 5 dB precision. The total ASSR elapsed time ranged from 60 to 90 min; if a response of low reliability was due to patient noise interference, then the subject was retested.
C-ABR and ASSR testing
The total number of ears tested was 151. Fifty five ears had no response to the C-ABR at the maximum presentation level of the device and so they were excluded from the correlation analysis; these ears were compared separately with the ASSR responses. The C-ABR and ASSR were recorded during the same test session, if possible, with the C-ABR testing always completed first. The data was excluded if the interval between the C-ABR and ASSR tests was greater than 2 months. The C-ABR thresholds were compared at 2 kHz, 4 kHz and the average of the 2 and 4 kHz ASSR thresholds, respectively. As a reference, the retrospectively assessed ASSR thresholds of 20 adults with normal hearing (9 men and 11 women, a total of 39 ears, mean age: 31.4 yr) were compared with those of 20 infants with normal hearing (11 boys and 9 girls, a total of 39 ears; mean age: 4.1 months) at carrier frequencies of 0.5, 1, 2, and 4 kHz.
RESULTS
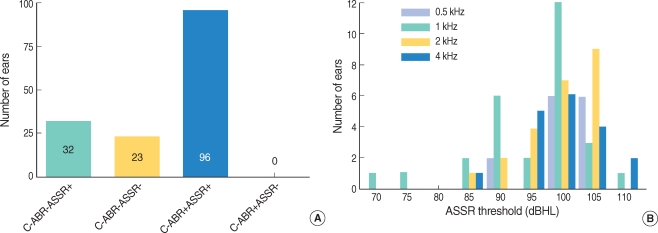
Of the 151 ears tested, 96 showed C-ABR responses, whereas 55 ears had no response to C-ABR at the maximum presentation level of the device. Of the latter 55 ears, 32 showed an ASSR response at 1 or more steady-state frequencies, whereas 23 ears showed the absence of both C-ABR and ASSR responses at all frequencies (Fig. 1A). There were no ears that responded to C-ABR testing, but not to ASSR testing.
The 32 ears showing an ASSR response, but no C-ABR response were analyzed in detail at frequencies of 90-110 dB HL (Fig. 1B).
When we analyzed the relationships between the thresholds obtained for the C-ABR and ASSR at 2 and 4 kHz and the average of the 2 and 4 kHz for all the tested subjects, there were significant correlations (r=0.92 for both the 2 and 4 kHz) between the high-frequency ASSR and C-ABR thresholds, with the average of the 2 and 4 kHz ASSR thresholds showing the highest correlation (r=0.94) with the C-ABR threshold (Fig. 2).
The differences between the adults and infants for the ASSR thresholds at frequencies of 0.5, 1, 2, and 4 kHz were compared. The ASSR thresholds of the 39 infant ears with normal hearing were elevated by an average of 8.5 dB compared with the thresholds of the 39 normal adult ears, with the differences being significant for all the frequencies. The adult subjects showed response threshold levels of approximately 21, 16, 15, and 16 dB HL for stimuli of 0.5, 1, 2, and 4 kHz frequency, respectively, whereas the thresholds obtained for the normal infants were somewhat higher. For the infants, the ASSR thresholds of approximately 33, 23, 23, and 23 dB HL were obtained at frequencies of 0.5, 1, 2, and 4 kHz, respectively. The differences in the averaged ASSR threshold between the adults and infants were 12, 7, 8, and 7 dB HL for 0.5, 1, 2, and 4 kHz, respectively (Table 1).
DISCUSSION
We have compared the 2 kHz, 4 kHz and the average of the 2 and 4 kHz ASSR thresholds with the C-ABR thresholds in infants in order to determine the effectiveness of the ASSR as a measure of hearing sensitivity in babies. We observed good correlations (r=0.92-0.94) between the C-ABR thresholds and the high frequency ASSR thresholds, and there was good agreement with the previously reported correlations (r=0.91-0.97) for infant subjects (2, 14, 15). These findings indicate that the ASSR is a promising frequency-specific test method.
The mean ASSR thresholds of the infants in this study ranged from 23 to 33 dB HL and they were about 10 dB HL higher than those of the normal hearing adults. Although there have been few direct comparisons because the subject groups and test parameters used for data collection and analysis varied slightly according to the clinic, many previous studies have reported similar results for infant subjects. For example, the ASSR thresholds in infants 1-10 months old were reported to be about 20-30 dB HL (16), and most other studies that focused on normal neonates and infants showed low-to-mid frequency levels of 35 to 45 dB HL and high frequency thresholds of 25 to 40 dB HL (17-19).
We found that the thresholds were 10 dB HL higher at low frequency than at high frequency, and this is in agreement with previous reported results that showed the ASSR thresholds in babies with normal hearing at 0.5 kHz were around 10 dB higher than those obtained for stimuli in the high frequency range (20). One hypothesis that may explain the higher threshold at low frequency than that at high frequency is that high frequency stimuli elicit relatively larger ASSR amplitudes in sedated subjects (12), which may lead to desynchronization of the neurons generating the responses because of jitter in the transmission time between the cochlear receptors and the neural generators (16). Another reason for the smaller responses to low frequency stimuli may be misplaced earphones, and this can greatly attenuate lower frequency stimuli. It is difficult to maintain earphones on a baby's ears at the proper position during the entire recording time (16). In addition, maturational changes may affect the transmission of low frequency energy in infants' middle ears (21).
The results obtained from 32 ears with an ASSR present at 1 or more steady-state frequencies when a C-ABR was absent at the maximum limitation of the device indicate that the ASSR thresholds can be used to predict hearing sensitivity for infants with severe-to-profound hearing loss. Several previous studies have been conducted to evaluate the advantages of ASSR (2, 14, 22).
CONCLUSION
The results of this study indicate that the ASSR test is objective, frequency-specific and well correlated with the C-ABR thresholds. The ASSR technique is of great advantage for subjects with no response at the maximum level of the C-ABR device. Moreover, the ASSR technique can be used for evaluating and managing pediatric subjects with sensorineural hearing loss ranging from severe to profound, and these patients require early intervention such as hearing aid fitting or a cochlear implant. The ASSR technique can supplement the C-ABR and extend the battery of pediatric tests that are available for assessing hearing impaired children.