|
 |
- Search
AbstractObjectivesInsertion of a silicone stent during endoscopic dacryocystorhinostomy (DCR) is the most common procedure to prevent rhinostomy closure. It has been claimed that silicone intubation improves the surgical outcomes of endoscopic DCR. However, many reports have documented an equally high success rate for surgery without silicone intubation. Accordingly, we conducted a systematic review and meta-analysis to clarify the outcomes of endoscopic DCR with and without silicone intubation and determine whether silicone intubation is actually beneficial for patients.
MethodsPubMed, Embase, and Cochrane Library databases were searched to identify relevant controlled trials evaluating endoscopic DCR with and without silicone intubation. The search was restricted to English articles published between January 2007 and December 2016. Relevant articles were reviewed to obtain information pertaining to interventions and outcomes. We also performed a meta-analysis of the relevant literature.
ResultsIn total, 1,216 patients included in 12 randomized controlled trials were pooled. A total of 1,239 endoscopic DCR procedures were performed, and silicone stents were used in 533 procedures. The overall success rate for endoscopic DCR was 91.9% (1,139/1,239), while the success rates with and without silicone intubation were 92.9% (495/533) and 91.2% (644/706), respectively. There was no statistically significant heterogeneity among the included studies. A meta-analysis using a fixed-effects models showed no significant difference in the success rate between endoscopic DCR with silicone intubation and that without silicone intubation (OR, 1.38; 95% CI, 0.89 to 2.12; P=0.148; z=1.45). Furthermore, there were no significant differences with regard to surgical complications such as synechia, granulation, and postoperative bleeding.
With the development of nasal endoscopes, endoscopic dacryocystorhinostomy (DCR) has become a popular and well-established procedure for patients with obstruction of the lacrimal system at the level of the sac or below [1]. Some of the common advantages of endoscopic DCR include the avoidance of an external scar; preservation of the pumping mechanism of the orbicularis muscle and avoidance of possible injury to the medial canthus; correction of associated intranasal pathologies such as deviated septum or rhinosinusitis; and a decreased surgical duration with better intraoperative visualization [2]. The reported success rate for endoscopic DCR ranges from 50% to 97% depending on the technique, obstruction level, and use of a silicone stent [3,4].
The most common reasons for the failure of endoscopic DCR include adhesions, restenosis, and obstruction of the common canaliculus [5-9]. Insertion of a silicone stent is the most common procedure to prevent closure of the rhinostomy [10-12]. It has been claimed that silicone intubation improves the surgical outcomes of endoscopic DCR by delaying fibrous closure during the postoperative healing period and consequently maintaining the patency of the fistula [13-17]. However, the use of silicone stenting during endoscopic DCR is a controversial topic. Some studies have reported that the silicone stent itself may cause tissue granulation, predisposing the site to postoperative infection, adhesions, and punctal lacerations and causing surgical failure [3,18-21]. Studies comparing endoscopic DCR with and without silicone intubation have been reported, and two meta-analyses concerning the benefits of silicone intubation during endoscopic DCR are available; however, the results are inconclusive [10,11].
The aim of this systematic review and meta-analysis was to clarify the surgical outcomes of endoscopic DCR with and without silicone intubation and determine whether silicone intubation is actually beneficial for patients.
We performed a systematic review of the published literature by searching PubMed, Embase, and Cochrane Library databases for English articles published between January 1, 2007 and December 31, 2016. The search was performed on July 13, 2017, and it was restricted to controlled trials comparing endoscopic DCR with and without silicone intubation. ‘‘Dacryocystorhinostomy’’ or ‘‘DCR” were used as the search terms. We read the titles of all articles and evaluated relevant abstracts. The full article was retrieved if the title, abstract, or both seemed to meet the objective of this review. The reference lists of original reports and review articles retrieved through the search were reviewed for additional studies that were not yet included in the computerized databases.
Two authors (HJJ and MGK) confirmed the inclusion of studies and mediated on the eligibility. Studies were only included if they compared an intervention group and a control group. Intervention was defined as the insertion of any form of silicone stent during endoscopic DCR for saccal or postsaccal obstruction. Control was defined as the noninsertion of a silicone stent. Other concomitant surgical procedures such as septoplasty or turbinectomy did not affect the eligibility of studies. Only randomized controlled trials (RCTs) were included in the final review; prospective cohort or retrospective cohort studies, noncomparative studies, and reviews were excluded. Studies were also excluded if the cohorts included cases of revision surgery or pediatric cases.
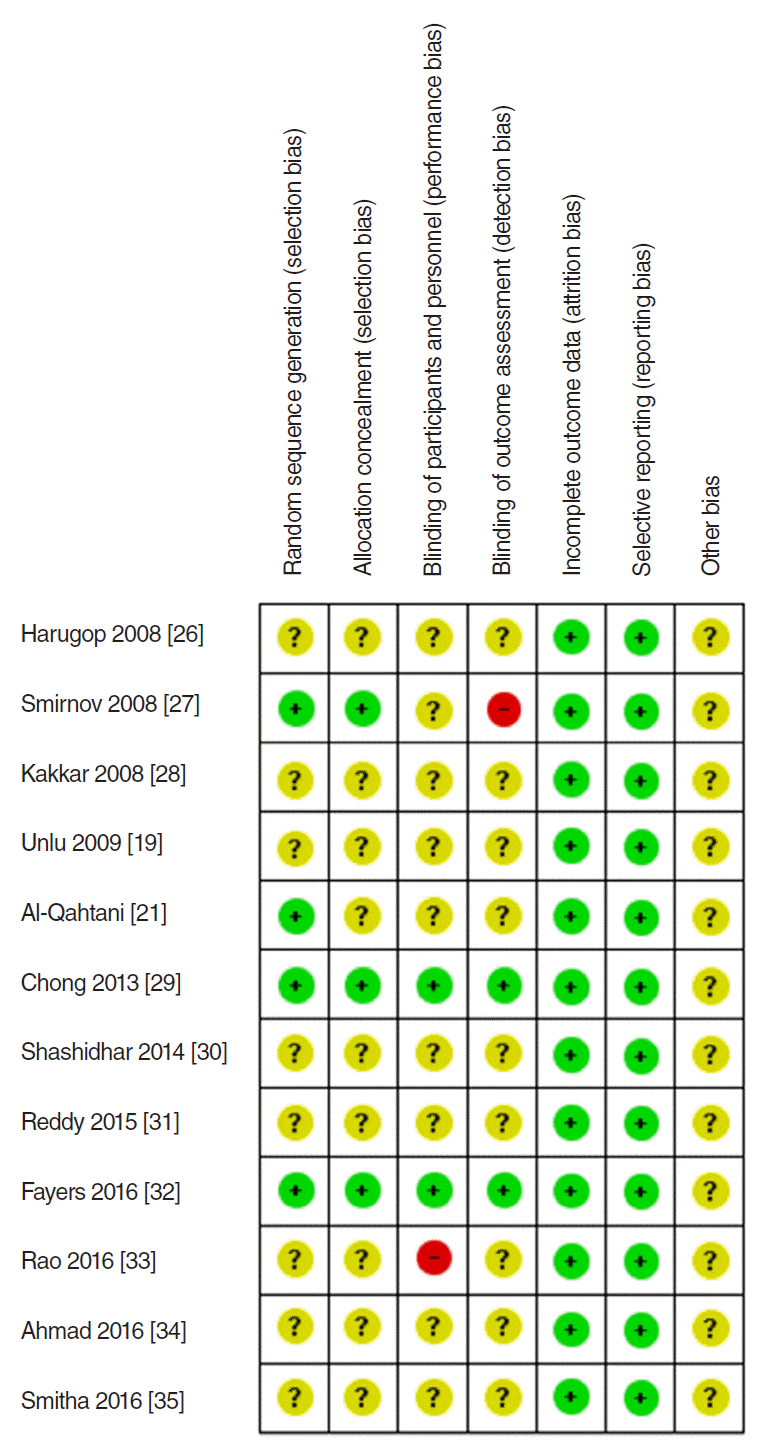
The authors independently assessed the sources of systematic bias in the trials according to the methods set out in the Cochrane Handbook for Systematic Reviews of Interventions [22]. The components of RCTs were as follows: random sequence generation, allocation concealment, blinding of participants, blinding of assessors, completeness of outcome data, selective reporting, and any other sources of bias.
Data were independently extracted by two reviewers according to a customized protocol. The following categories of information were extracted: study characteristics, including the author, publication year, and country of publication; and patient characteristics, including age, sex, type of surgical intervention, duration of follow-up, and outcome measures.
The primary outcome was surgical success, which was measured subjectively and objectively. Adequate resolution of related symptoms was the eligible subjective measure, while endoscopic visualization of the rhinostomy opening, a positive functional endoscopic dye test, and positive lacrimal syringing were the eligible objective measures. If the surgical success rate was separately reported for objective and subjective assessments, and if the overall surgical success rate could not be extracted, the subjective surgical success rate was used for analysis. Studies with no data regarding success as defined above were excluded, as were those reporting a symptom scale or score improvement without an absolute improvement. Adverse events such as postoperative bleeding, scarring, synechia, and granulation were assessed as secondary outcome measures.
The meta-analysis was performed using R software package ver. 3.3.1 (R Foundation, Vienna, Austria). The outcomes are presented as pooled odds ratio (OR) with 95% confidence interval (CI). The weight of the individual studies involved in the meta-analysis was calculated using the Mantel-Haenszel method [23]. Statistical heterogeneity was assessed using a chi-square equation. The I2-statistic was calculated to analyze the between-study heterogeneity. A fixed-effects model was used if the I2-statistic was <0.4. The meta-analyzed data are demonstrated as forest plots generated using the “meta” packages (ver. 4.9) of R software [24]. An additional meta-analysis was performed to compare the postoperative complication rate between DCR with and without silicone intubation. The Harbord test was used to assess publication bias for effects measured by ORs and displayed using funnel plots [25]. A P-value <0.05 was considered statistically significant.
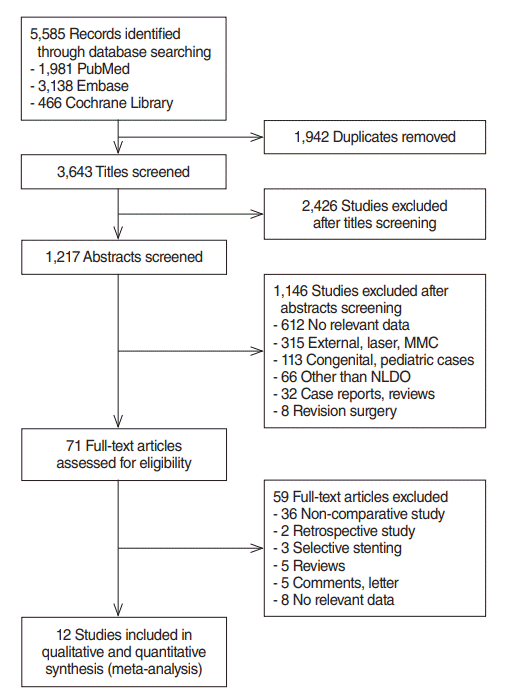
Fig. 1 shows our search results. In total, 5,585 citations (1,981 from PubMed, 3,138 from Embase, and 466 from the Cochrane Library) were identified; the number was brought down to 1,217 after initial title screening as per the inclusion and exclusion criteria. Studies involving cases of external DCR, laser-assisted DCR, and mitomycin-C applied DCR; pediatric cases; and cases of revision surgery were excluded, as were review articles and case reports. The full-text of 71 articles were reviewed, and 36 noncomparative studies and five nonrandomized studies were excluded. Eventually, 12 RCTs were considered eligible for analysis [19,21,26-35]. The characteristics of the eligible studies are summarized in Table 1. Each included study was assessed for the risk of bias using the Cochrane Collaboration tool (Fig. 2).
A total of 1,239 procedures were performed in 1,216 patients included in the 12 enrolled studies. All studies recruited patients with either saccal or postsaccal obstruction who underwent diagnostic assessments with various combinations of lacrimal probing, lacrimal syringing, nasal endoscopy, Jones I and II tests, and dacryocystography. All patients were aged ≥17 years, with a mean age of 33.8 to 64.0 years. The duration of follow-up before the assessment of surgical success was 2.5 months in one study; the mean follow-up duration in the remaining studies was 17.4 months (range, 6 to 97 months). The average extubation time ranged from 1.5 to 4 months. Concomitant interventions, including septoplasty and turbinoplasty, were performed in six studies. After surgery, patients were discharged with antibiotics alone or in combination with intranasal steroids and saline nasal sprays or steroid eyedrops and xylometazoline nasal spray. Although exclusion criteria varied among studies, common ones included previous sinonasal malignancies, previous lacrimal malignancies, and previous lacrimal surgery.
All studies had explicitly defined the criteria for success, which were usually a combination of subjective symptomatic improvement and the objective confirmation of anatomical patency of the nasolacrimal system. The most common subjective criterion was gross symptom improvement. Four different objective measures were used. These included one or more of the following, in decreasing order of frequency: irrigation of the nasolacrimal system, endoscopic inspection of the stoma, the functional endoscopic dye test, and the Jones I test.
The success rate for endoscopic DCR in the 12 included studies ranged from 85.0% to 96.2%. The overall success rate was 91.9% (1,139/1,239). Bicanalicular silicone stents were used in 533 procedures; the remaining 644 procedures were performed without silicone stenting. The success rates for endoscopic DCR with and without silicone intubation were 92.9% (495/533) and 91.2% (644/706), respectively. Heterogeneity testing showed that there was no significant heterogeneity among studies (Q=10.61, df=11, I2=0%, P=0.48). According to prespecified criteria for heterogeneity, a fixed-effects model was used for subsequent analysis.
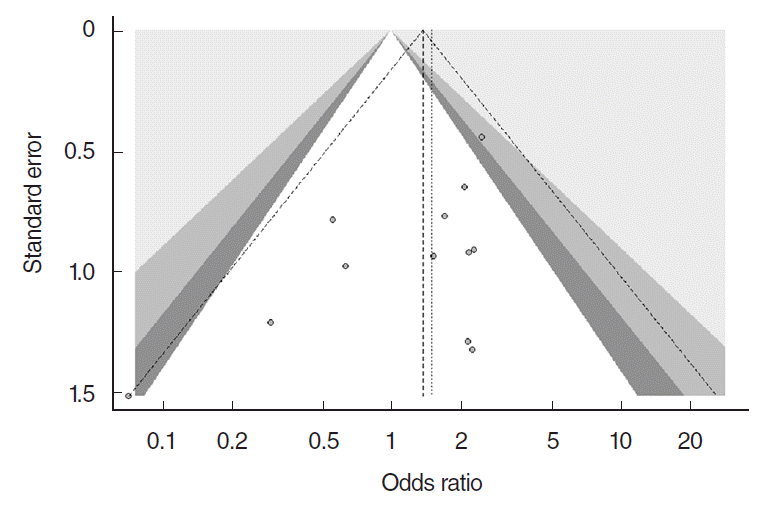
Our meta-analysis revealed that there was no significant difference in the surgical success rate between endoscopic DCR with silicone intubation and that without silicone intubation (OR, 1.38; 95% CI, 0.89 to 2.12; P=0.148; z=1.45) (Fig. 3). Publication bias was depicted by funnel plots, and there was no evidence of publication bias (t=−1.677, df=10, P=0.124 by the Harbord test) (Fig. 4).
Of the 12 RCTs, 11 reported postoperative complications, including granulation tissue formation, synechia, adhesion, postoperative bleeding, and other complications associated with the silicone tube, such as discomfort from stenting, punctal laceration, stent extrusion or prolapse, and difficulty in stent removal. The most commonly reported postoperative complications were synechia between the middle turbinate and nasal lateral wall and granulation tissue formation around the stoma. Synechia, granulation tissue formation, and postoperative bleeding were reported in five (Fig. 5), four (Fig. 6), and three (Fig. 7) studies, respectively. All the forest plots revealed no significant differences in the surgical complication rate between endoscopic DCR with silicone intubation and that without silicone intubation. The reported complications considered to be associated with the silicone stent itself are summarized in Table 2.
Through the present systematic review and meta-analysis, we found that there were no significant differences between endoscopic DCR with silicone intubation and that without silicone intubation with regard to the surgical success rate and the postoperative complication rate.
Silicone stenting was first introduced in the 1950s for external DCR and the 1960s for endoscopic DCR [32,36]. Thereafter, stents rapidly became favored by surgeons who perform lacrimal surgery [37]. However, there is some controversy regarding the use of stenting during endoscopic DCR. Those who advocate its use report an increased postoperative patency rate associated with maintenance of the opening of the ostium [15,16]. However, other studies reported a higher failure rate with silicone intubation because of granulomatous inflammation, canalicular injury, and patient discomfort [27,37-39]. An increased surgical duration, increased postoperative care, and high surgical costs have been cited as additional disadvantages of stenting [21]. In addition, a number of studies have reported satisfactory success rates ranging from 87% to 93% for endoscopic DCR without stent intubation [39-43].
Therefore, several studies have attempted to compare endoscopic DCR with and without stent intubation and determine the technique with the maximum clinical benefits. A multicenter, randomized, prospective interventional trial of stenting versus nonstenting in patients undergoing endoscopic DCR could provide the answer to the question; however, all previous RCTs had very small sample sizes. A meta-analysis that included two RCTs showed no advantage of the use of silicone intubation [10]. Another study also showed no benefit of silicone intubation with regard to the success rate for endoscopic DCR, with poor evidence to support the use of silicone stenting for improved surgical outcomes [11]. However, the two available meta-analyses included only two and five RCTs, respectively, and did not include recently published RCTs [10,11]. In addition, a recent study showed the meta-analysis results by subgroup analysis with six RCTs that there was no significant difference in endoscopic DCR group [44]. Funnel plots are commonly used to evaluate publication bias, and Sterne et al. [45] recommended that a test for funnel plot asymmetry should be conducted only if the number of studies included in a meta-analysis is ≥10. Therefore, the previous meta-analyses concerning endoscopic DCR have critical limitations with regard to their statistical methods. Our meta-analysis has some distinct advantages over the previous studies because it included 12 of the most recent RCTs, which was sufficient for the construction of funnel plots for the evaluation of publication bias.
Our meta-analysis showed that the success rate for endoscopic DCR was not affected by the use of silicone intubation. We also calculated sequential cumulative meta-analyzed results for each year from 2007 and found that the success rate for endoscopic DCR with silicone intubation improved from 2012 onwards. Although there was a tendency to prefer the use of silicone intubation during endoscopic DCR, the trend was not statistically significant (Fig. 8).
Common complications after endoscopic DCR included synechia, granulation tissue formation, postoperative bleeding, and other complications associated with the silicone tube, including discomfort from stenting, stent extrusion, difficulty in stent removal, and punctal laceration. Because many studies did not provide quantitative values for surgical complications, only a small number of studies was analyzed for surgical complications. Our meta-analysis revealed that there was no significant difference in the surgical complication rate between endoscopic DCR with silicone intubation and that without silicone intubation, suggesting that the use of silicone stents does not increase the risk of synechia, granulation tissue formation, and postoperative bleeding.
As pointed out in a recent systematic review [2], a meta-analysis of various studies on the benefits of stent intubation during endoscopic DCR is extremely difficult because of wide variations in patient selection procedures, outcome definitions (subjective vs. objective), and surgical technique. In addition, in the absence of unified preoperative examinations, many studies only used probing and irrigation to determine the obstruction level, which is an important factor influencing postoperative outcomes. Other factors such as the use of a mucosal flap also influence the surgical outcome. Since the concept of flaps has become popular in recent years, we only included studies published within the last 10 years. Mucosal flaps are reflected to protect the surrounding bone and avoid scarring and synechia [38]. Mucosal preservation and the avoidance of bone exposure cannot be overemphasized and are crucial for optimal results [46]. More so, there is no general agreement regarding the extubation time [5]. The optimal duration of silicone stenting is controversial, with the reported duration ranging from 4 weeks to 4 months [21,47]. In the present study, the extubation time was 1.5–4 months. In addition, there are many variations, without any standard, in the outcome measures among studies. The variables discussed above can all influence the success rate, which makes it difficult to conduct a well-controlled study and meta-analysis. The strength of this study is that it derives its results from a meta-analysis of the largest number of RCTs till date (12 RCTs published in the past 10 years), which were carefully selected to be as similar as possible to each other.
In conclusion, the findings of our meta-analysis suggest that the success rate and complication rate for endoscopic DCR is not influenced by the use of silicone intubation during the procedure. There is no clear evidence showing that stent intubation during endoscopic DCR is superior to nonintubation.
▪ There is some controversy regarding the use of silicone stenting in endoscopic dacryocystorhinostomy.
▪ Meta-analysis showed no significant differences with regard to the surgical success rate and the postoperative complications.
▪ There was no clear evidence showing that stent intubation during endoscopic dacryocystorhinostomy is superior to nonintubation.
Fig. 3.Comparison of success rate between endoscopic dacryocystorhinostomy with and without silicone intubation. OR, odds ratio; CI, confidence interval. 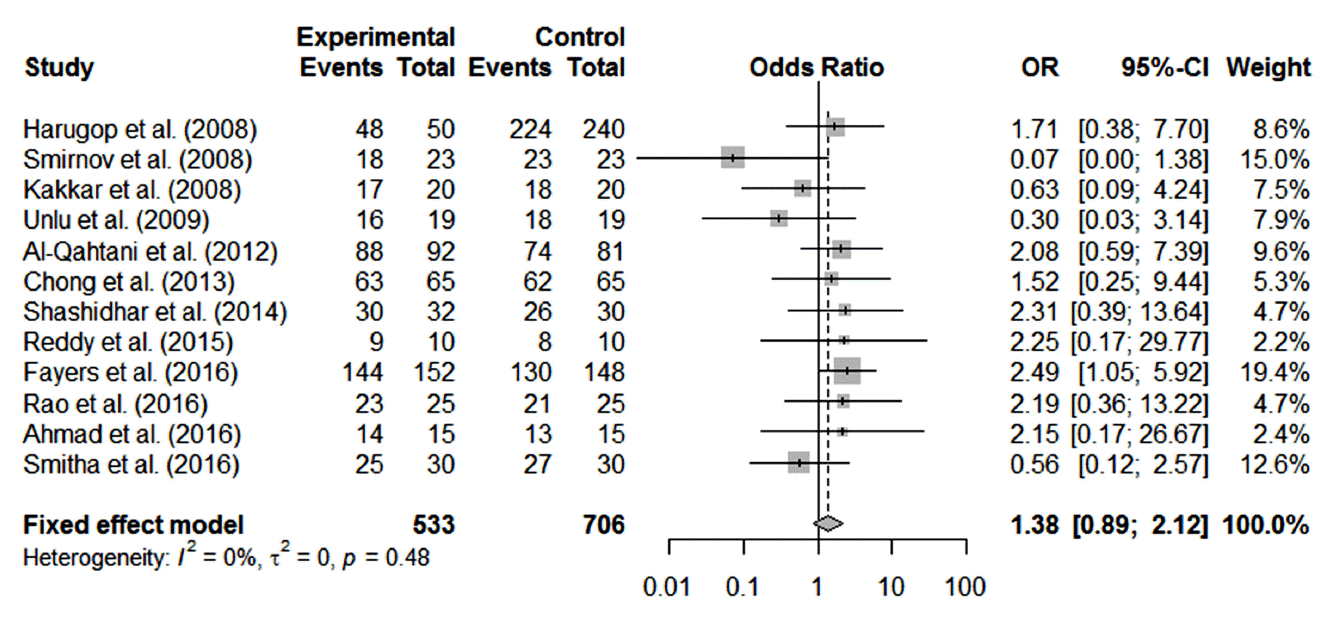
Fig. 5.Postoperative complication (synechia) of endoscopic dacryocystorhinostomy with and without silicone intubation. OR, odds ratio; CI, confidence interval. 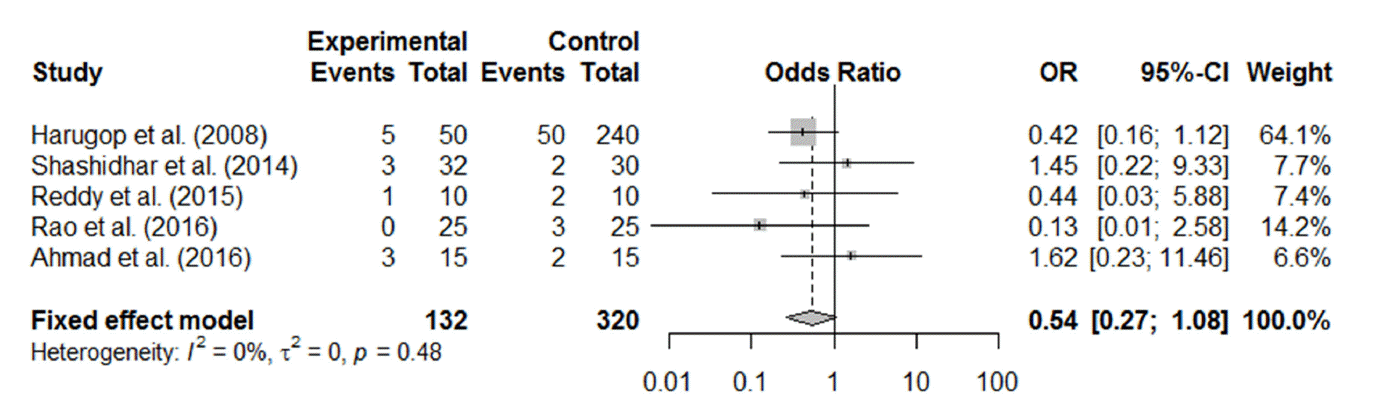
Fig. 6.Postoperative complication (granulation) of endoscopic dacryocystorhinostomy with and without silicone intubation. OR, odds ratio; CI, confidence interval. 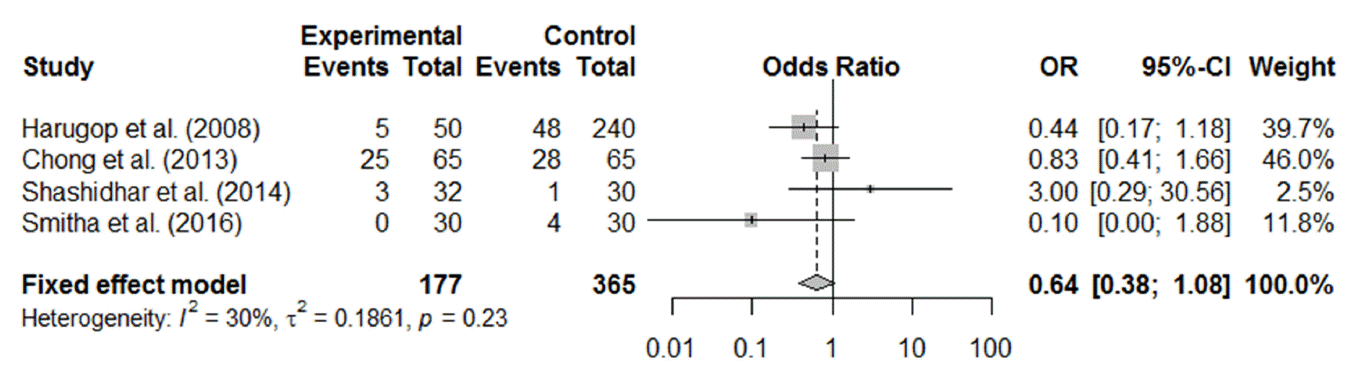
Fig. 7.Postoperative complication (postoperative bleeding) of endoscopic dacryocystorhinostomy with and without silicone intubation. OR, odds ratio; CI, confidence interval. 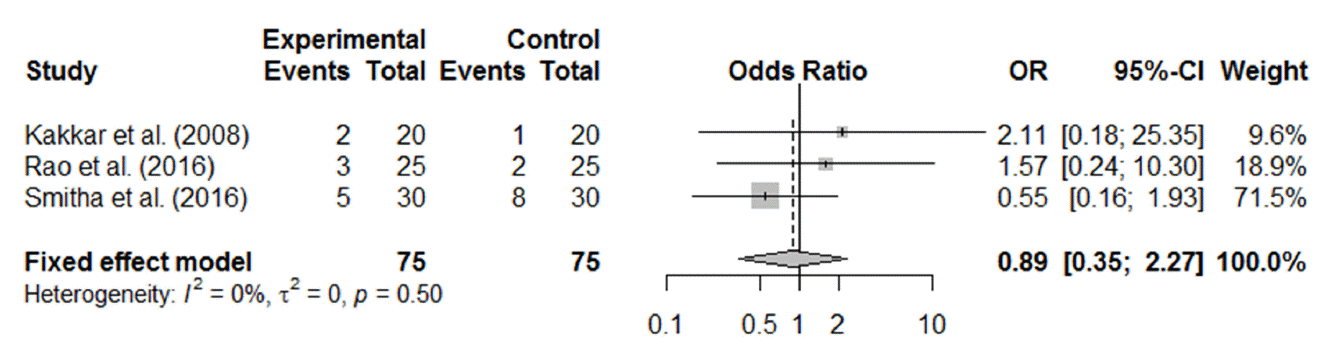
Fig. 8.Sequential cumulative meta-analysis results of 12 included study. OR, odds ratio; CI, confidence interval. 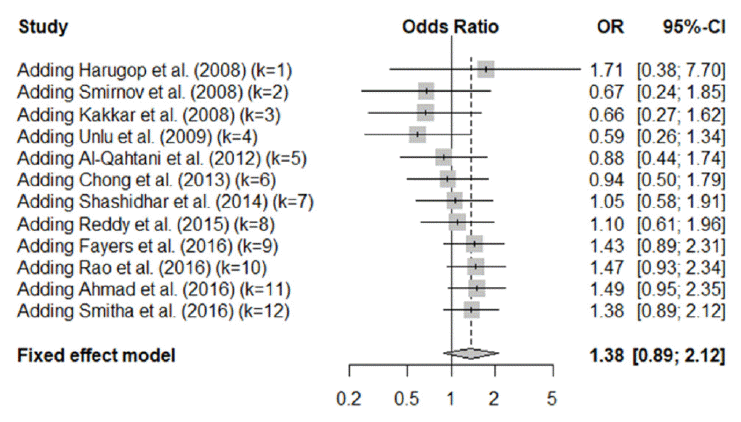
Table 1.Characteristics of the included studies
Table 2.Complications related to silicone intubation
REFERENCES1. Unlu HH, Toprak B, Aslan A, Guler C. Comparison of surgical outcomes in primary endoscopic dacryocystorhinostomy with and without silicone intubation. Ann Otol Rhinol Laryngol. 2002 Aug;111(8):704-9.
2. Leong SC, Macewen CJ, White PS. A systematic review of outcomes after dacryocystorhinostomy in adults. Am J Rhinol Allergy. 2010 Jan-Feb;24(1):81-90.
3. Feng YF, Cai JQ, Zhang JY, Han XH. A meta-analysis of primary dacryocystorhinostomy with and without silicone intubation. Can J Ophthalmol. 2011 Dec;46(6):521-7.
4. Callejas CA, Tewfik MA, Wormald PJ. Powered endoscopic dacryocystorhinostomy with selective stenting. Laryngoscope. 2010 Jul;120(7):1449-52.
5. Syed MI, Head EJ, Madurska M, Hendry J, Erikitola OC, Cain AJ. Endoscopic primary dacryocystorhinostomy: are silicone tubes needed? Our experience in sixty three patients. Clin Otolaryngol. 2013 Oct;38(5):406-10.
6. McLachlan DL, Shannon GM, Flanagan JC. Results of dacryocystorhinostomy: analysis of the reoperations. Ophthalmic Surg. 1980 Jul;11(7):427-30.
7. Sprekelsen MB, Barberan MT. Endoscopic dacryocystorhinostomy: surgical technique and results. Laryngoscope. 1996 Feb;106(2 Pt 1):187-9.
8. Boush GA, Lemke BN, Dortzbach RK. Results of endonasal laser-assisted dacryocystorhinostomy. Ophthalmology. 1994 May;101(5):955-9.
9. Watkins LM, Janfaza P, Rubin PA. The evolution of endonasal dacryocystorhinostomy. Surv Ophthalmol. 2003 Jan-Feb;48(1):73-84.
10. Gu Z, Cao Z. Silicone intubation and endoscopic dacryocystorhinostomy: a meta-analysis. J Otolaryngol Head Neck Surg. 2010 Dec;39(6):710-3.
11. Sarode D, Bari DA, Cain AC, Syed MI, Williams AT. The benefit of silicone stents in primary endonasal dacryocystorhinostomy: a systematic review and meta-analysis. Clin Otolaryngol. 2017 Apr;42(2):307-14.
12. Okuyucu S, Gorur H, Oksuz H, Akoglu E. Endoscopic dacryocystorhinostomy with silicone, polypropylene, and T-tube stents; randomized controlled trial of efficacy and safety. Am J Rhinol Allergy. 2015 Jan-Feb;29(1):63-8.
13. Tsirbas A, Wormald PJ. Endonasal dacryocystorhinostomy with mucosal flaps. Am J Ophthalmol. 2003 Jan;135(1):76-83.
14. Onerci M, Orhan M, Ogretmenoglu O, Irkec M. Long-term results and reasons for failure of intranasal endoscopic dacryocystorhinostomy. Acta Otolaryngol. 2000 Mar;120(2):319-22.
15. Metson R, Woog JJ, Puliafito CA. Endoscopic laser dacryocystorhinostomy. Laryngoscope. 1994 Mar;104(3 Pt 1):269-74.
16. Griffiths JD. Nasal catheter use in dacryocystorhinostomy. Ophthal Plast Reconstr Surg. 1991 Sep;7(3):177-86.
17. Caversaccio M, Hausler R. Insertion of double bicanalicular silicone tubes after endonasal dacryocystorhinostomy in lacrimal canalicular stenosis: a 10-year experience. ORL J Otorhinolaryngol Relat Spec. 2006 Aug;68(5):266-9.
18. Saeed BM. Endoscopic DCR without stents: clinical guidelines and procedure. Eur Arch Otorhinolaryngol. 2012 Feb;269(2):545-9.
19. Unlu HH, Gunhan K, Baser EF, Songu M. Long-term results in endoscopic dacryocystorhinostomy: is intubation really required. Otolaryngol Head Neck Surg. 2009 Apr;140(4):589-95.
20. Allen K, Berlin AJ. Dacryocystorhinostomy failure: association with nasolacrimal silicone intubation. Ophthalmic Surg. 1989 Jul;20(7):486-9.
21. Al-Qahtani AS. Primary endoscopic dacryocystorhinostomy with or without silicone tubing: a prospective randomized study. Am J Rhinol Allergy. 2012 Jul-Aug;26(4):332-4.
22. Higgins JP, Green S. Cochrane handbook for systematic reviews of interventions version 5.1.0 [Internet]. London: The Cochrane Collaboration; 2011 [cited 2018 Feb 8]. Available from: http://www.handbook.cochrane.org.
23. Mantel N, Haenszel W. Statistical aspects of the analysis of data from retrospective studies of disease. J Natl Cancer Inst. 1959 Apr;22(4):719-48.
24. Schwarzer G. General package for meta-analysis [Internet]. 2018 [cited 2017 Dec 07]. Available from: http://cran.r-project.org/web/packages/meta/meta.pdf.
25. Harbord RM, Deeks JJ, Egger M, Whiting P, Sterne JA. A unification of models for meta-analysis of diagnostic accuracy studies. Biostatistics. 2007 Apr;8(2):239-51.
26. Harugop AS, Mudhol RS, Rekha BK, Maheswaran M. Endonasal dacryocystorhinostomy: a prospective study. Indian J Otolaryngol Head Neck Surg. 2008 Dec;60(4):335-40.
27. Smirnov G, Tuomilehto H, Terasvirta M, Nuutinen J, Seppa J. Silicone tubing is not necessary after primary endoscopic dacryocystorhinostomy: a prospective randomized study. Am J Rhinol. 2008 Mar-Apr;22(2):214-7.
28. Kakkar V, Chugh J, Sachdeva S, Sharma N, Ramesh. Endoscopic dacryocystorhinostomy with and without silicone stent: a comparative study. Internet J Otorhinolaryngol. 2008;9(1):1-5.
29. Chong KK, Lai FH, Ho M, Luk A, Wong BW, Young A. Randomized trial on silicone intubation in endoscopic mechanical dacryocystorhinostomy (SEND) for primary nasolacrimal duct obstruction. Ophthalmology. 2013 Oct;120(10):2139-45.
30. Shashidhar KC, Nagalotimath US, Dixit D. Endoscopic dacryocystorhinostomy with and without silicone stenting: a comparative study. Al Ameen J Med Sci. 2014 Jul-Sep;7(3):244-7.
31. Reddy YJ, Reddy YM, Kiran M, Reddy YG, Kumar S. A comparative study of outcomes of dacryocystorhinostomy with and without silicone stenting. IOSR J Dent Med Sci. 2015 Jul;14(7):82-5.
32. Fayers T, Dolman PJ. Bicanalicular silicone stents in endonasal dacryocystorhinostomy: results of a randomized clinical trial. Ophthalmology. 2016 Oct;123(10):2255-9.
33. Rao SV, Rajshekar MM. Dacryocystorhinostomy stent insertion in initial endoscopic dacryocystorhinostomy. Clin Rhinol An Int J. 2016 Sep-Dec;9(3):120-4.
34. Ahmad S, Pant B. Role of silicone stenting in endoscopic dacryocystorhinostomy: a comparative study. Int J Adv Integr Med Sci. 2016 Jan-Mar;1(1):4-6.
35. Smitha SG, Jagannath B. Endoscopic dacryocystorhinostomy with and without silicone stent: a comparative study. J Med Sci Clin Res. 2016 Sep;4(9):12861-4.
36. Madge SN, Selva D. Intubation in routine dacryocystorhinostomy: why we do what we do. Clin Exp Ophthalmol. 2009 Aug;37(6):620-3.
37. Liang J, Lane A. Is postoperative stenting necessary in endoscopic dacryocystorhinostomy. Laryngoscope. 2013 Nov;123(11):2589-90.
38. Longari F, Dehgani Mobaraki P, Ricci AL, Lapenna R, Cagini C, Ricci G. Endoscopic dacryocystorhinostomy with and without silicone intubation: 4 years retrospective study. Eur Arch Otorhinolaryngol. 2016 Aug;273(8):2079-84.
39. Mohamad SH, Khan I, Shakeel M, Nandapalan V. Long-term results of endonasal dacryocystorhinostomy with and without stenting. Ann R Coll Surg Engl. 2013 Apr;95(3):196-9.
40. Mortimore S, Banhegy GY, Lancaster JL, Karkanevatos A. Endoscopic dacryocystorhinostomy without silicone stenting. J R Coll Surg Edinb. 1999 Dec;44(6):371-3.
41. Unlu HH, Ozturk F, Mutlu C, Ilker SS, Tarhan S. Endoscopic dacryocystorhinostomy without stents. Auris Nasus Larynx. 2000 Jan;27(1):65-71.
42. Zuercher B, Tritten JJ, Friedrich JP, Monnier P. Analysis of functional and anatomic success following endonasal dacryocystorhinostomy. Ann Otol Rhinol Laryngol. 2011 Apr;120(4):231-8.
43. Gibbs DC. New probe for the intubation of lacrimal canaliculi with silicone rubber tubing. Br J Ophthalmol. 1967 Mar;51(3):198.
44. Deng HY, Wang T, Huang XK, Yang QT, Ling SQ, Wang WH, et al. Comparative study of recessive spherical headed silicone intubation and endonasal dacryocystorhinostomy under nasal endoscopy for nasolacrimal duct obstruction. Sci Rep. 2017 Aug;7(1):7734.
45. Sterne JA, Sutton AJ, Ioannidis JP, Terrin N, Jones DR, Lau J, et al. Recommendations for examining and interpreting funnel plot asymmetry in meta-analyses of randomized controlled trials. BMJ. 2011 Jul;343:d4002.
|
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||