

AUTHOR CONTRIBUTIONS
Conceptualization: SHH. Data curation: GS, YJK. Formal analysis: SHH. Funding acquisition: SHH, YJK. Methodology: SHH, GS. Project administration: SHH. Visualization: SHH. Writing–original draft: YJK, SHH. Writing–review & editing: YJK, GS.
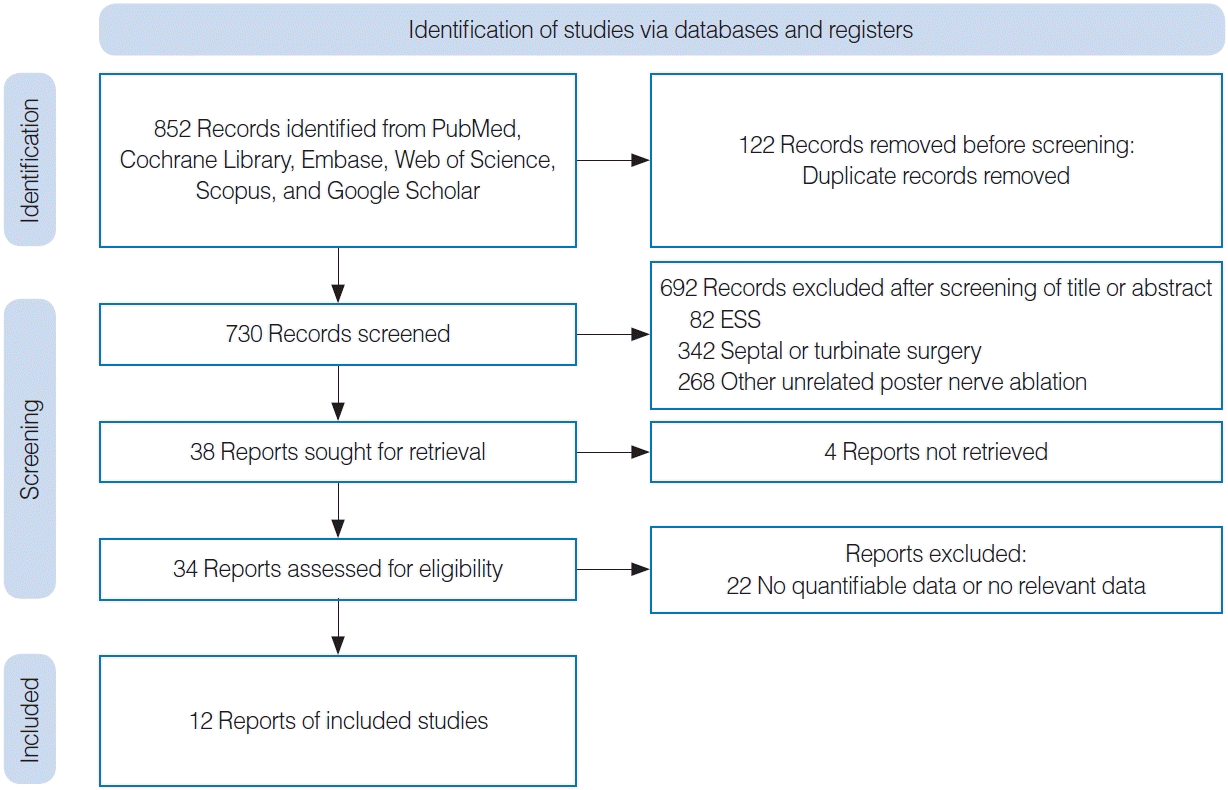


| Study | Design | Total number of patients | Age of patients (yr, mean±SD) | Sex (male:female) | Nonallergic rhinitis:allergic rhinitis number of patients (n) | Nation | Treatment device | Outcome |
|---|---|---|---|---|---|---|---|---|
| Hwang et al. (2017) [12] | Prospective observational study | 27 | 53.3±3.3 | 6:17 | 14:13 | USA | ClariFix device | Total TNSS |
| Chang et al. (2020) [10] | Prospective, single-arm study | 98 | 58.6±16.2 | 35:63 | 70:28 | USA | ClariFix device | RQLQ, total TNSS, response rate (TNSS) |
| Yen et al. (2020) [11] | Prospective, nonrandomized, interventional, post-market feasibility study | 30 | 60.0±15.8 | 14:16 | 17:11 | USA | ClariFix device | RQLQ, total TNSS |
| Stolovitzky et al. (2021) [15] | Prospective, single-blinded, randomized controlled, multicenter study | 117 | 57.3±14.8 | 42:75 | NA | USA | RhinAer System | Total TNSS |
| Ow et al. (2021) [9] | Prospective, single-arm, multicenter study | 62 | 57.1±13.4 | 22:40 | 43:19 | USA | ClariFix device | Total TNSS |
| Gerka Stuyt et al. (2021) [18] | Prospective, single-arm study | 24 | 60.04±16.7 | 12:12 | 16:8 | USA | ClariFix device | Total TNSS |
| Ehmer et al. (2022) [20] | Prospective, single-arm, multicenter study | 50 | 57.9±11.9 | 21:29 | 17:21 (12 Unknown patients) | USA | RhinAer System | Total TNSS, response rate (TNSS) |
| Lee et al. (2022) [13] | Prospective, single-arm study | 126 | 57.9±13.4 | 60:66 | 93:10 (1 Mixed rhinitis patient, 25 unknown patients) | USA | RhinAer System | RQLQ, total TNSS, response rate (RQLQ), response rate (TNSS) |
| Takashima et al. (2023) [19] | Prospective, patient-blinded, randomized controlled, multicenter study | 77 | 57.3±14.8 | 28:49 | NA | USA | RhinAer System | Total TNSS, response rate (TNSS) |
| Del Signore et al. (2022) [8] | Prospective, patient-blinded, randomized, sham-controlled, multicenter study | 127 | 55.2±16.3 | 77:56 | 76:57 | USA | ClariFix device | RQLQ, total TNSS, response rate (RQLQ), response rate (TNSS) |
| Reh et al. (2023) [6] | Prospective, single-arm, multicenter study | 36 | 62.8±14.0 | 17:19 | 13:23 | USA | NEUROMARK System | Total TNSS, response rate (TNSS) |
| Virani et al. (2023) [4] | Retrospective study | 14 | 59.1±14.8 | 8:6 | 10:4 | USA | Cryotherapy | RQLQ, total TNSS |
| Group |
Follow-up |
Rate of good responsea) | Rate of clinical responseb) | ||||
|---|---|---|---|---|---|---|---|
| 1 mo | 3 mo | 6 mo | 12 mo | 24 mo | |||
| Cryotherapy | (n=5) | (n=6) | (n=2) | (n=3) | (n=1) | (n=2) | (n=2) |
| MD, 3.5265; 95% CI, 3.3346–3.7185; I2=0.0% | MD, 3.5026; 95% CI, 3.2989–3.7063; I2=0.0% | MD, 3.5388; 95% CI, 2.7585–4.3191; I2=83.7% | MD, 3.7584; 95% CI, 2.8071–4.7096; I2=85.6% | MD, 4.1000; 95% CI, 3.3362–4.8638; I2=NA | 73.7%; 95% CI, 0.6639–0.7998; I2=0.0% | 81.8%; 95% CI, 0.7393–0.8772; I2=0.0% | |
| Radiofrequency ablation | (n=2) | (n=4) | (n=3) | (n=3) | (n=1) | (n=4) | (n=4) |
| MD, 4.2627; 95% CI, 2.9794–5.5460; I2=79.0% | MD, 4.1399; 95% CI, 3.5476–4.7322; I2=67.3% | MD, 4.8121; 95% CI, 4.4057–5.2185; I2=27.5% | MD, 4.8740; 95% CI, 4.3979–5.3500; I2=0.0% | MD, 5.5000; 95% CI, 4.6863– 6.3137; I2=NA | 76.9%; 95% CI, 0.6913–0.8311; I2=41.3% | 91.9%; 95% CI, 0.8788–0.9465; I2=0.0% | |
| P-value | 0.266 | 0.046 | 0.005 | 0.040 | 0.014 | 0.536 | 0.005 |