INTRODUCTION
The recent expansion of the U.S. Food and Drug Administration (FDA) criteria for cochlear implantation (CI) to include infants as young as 9 months [
1,
2] has reignited long-standing debates about the optimal age for pediatric CI. A particularly recent retrospective study from the United States showed that children who received CI before 9 months of age exhibited superior receptive and expressive language development after an average follow-up of 7.4 years, compared to those who received CI between 9 and 12 months [
3]. There is a growing body of evidence supporting the lasting benefits of CI at increasingly younger ages [
4-
7], and little research suggests that the advantages of early CI diminish when performed before 9 months [
3,
8-
10]. We recommend fitting hearing aids by no later than 6 months of age, or even earlier for newborns with significant hearing loss, in accordance with the Joint Committee on Infant Hearing (JCIH) guidelines, which recommend timely intervention by 6 months of age. Extending this rationale, it may be beneficial, or even essential, for infants with profound, irreversible sensorineural hearing loss to receive CI surgery before reaching 9 months of age. Such an approach is in line with the JCIH’s recommendations for early intervention and has the potential to maximize the benefits for language development.
However, while hearing aids are widely accepted, there are concerns regarding CI at a very early age, such as before 9 months. These concerns include the potential side effects of general anesthesia in infants, perioperative risks associated with such an early age, and the difficulties in accurately assessing hearing status in very young infants. However, recent reports suggest that the side effects of general anesthesia and surgery in very young children are not considered significant issues [
7,
11]. Nevertheless, before routinely implementing CI in infants, several issues need further clarification. First, the existing literature does not provide adequate information about the causes of hearing loss or the degree of speech development delay prior to CI in the very early CI group, such as those younger than 9 months, compared to those who receive the implant later. Most studies have focused on speech development outcomes after surgery, without offering detailed accounts of pre-CI language development or the criteria for performing CI before 9 months or between 9 and 12 months of age. Second, it is unclear whether the benefits of very early CI are immediately apparent, even within the first year following the procedure, in terms of receptive and expressive language development. Although children who received their implants and activation before 9 months of age have been reported to exhibit rapid auditory skill development, reaching the expected performance level of normal-hearing peers by the age of 2 [
10], the early trajectory of receptive and expressive language development in the first 1–2 years post-implantation for the under-9-months group—which could reflect the direct impact of CI without confounding factors—compared to long-term outcomes, remains largely unexplored.
Given this, we aim to provide comparative data on the early speech development of children who received CI before 9 months of age versus those who received it between 9 and 12 months. Our pediatric cohort consisted of cases where the genetic and radiologic etiology was clearly documented in over 90% of instances. Furthermore, the decision regarding the timing of CI is based solely on objective criteria that focus on the extent of language development delay compared to age-matched controls. By adopting this methodology, we strive to minimize the potential influence of the infant’s parents’ socioeconomic status (SES) or their enthusiasm for auditory rehabilitation on the timing of CI. The South Korean healthcare insurance system has played a significant role in facilitating this approach. This method should enable us to accurately assess the impact of the age at CI on language development during the first 1–2 years postoperatively.
DISCUSSION
Children with congenital severe to profound hearing loss are now being identified at increasingly younger ages, a trend likely linked to the global, gradual implementation of newborn hearing screening programs. This earlier detection naturally results in the earlier fitting of hearing aids and is anticipated to lower the age at which CI is performed. Numerous studies have demonstrated that performing CI on children with profound hearing loss before the age of 12 months is as safe as CI at later ages. It does not result in a higher rate of complications and is more efficacious [
28-
31].
The gradual and prolonged reduction of the minimum age for pediatric CI by the U.S. FDA—from 12 months to 9 months—over a span of 20 years underscores the complexity of this decision, given the rigorous evidence required. Factors that have influenced the move toward earlier CI include early fitting of hearing aids, confirmed profound hearing loss, higher SES, and the presence of specific genetic markers such as the
GJB2 variant [
32]. Financial and rehabilitative commitments, especially in countries lacking governmental support for CI, often restrict access to the procedure to more affluent families. Additionally, the challenge of obtaining reliable auditory responses from very young infants adds complexity to early CI decisions. This is further complicated by the uncertainty surrounding the etiology of hearing loss, which can lead to hesitancy among guardians and surgeons. A more accurate evaluation of the effects of very early CI would be best achieved through a cohort study designed to minimize biases and hesitancy.
Our center employs a comprehensive set of criteria to determine the appropriate timing for CI in pre-lingual pediatric candidates. Initially, we conduct genetic testing and developmental assessments, including the BSID-4, and rigorously evaluate the progress of language development with the aid of hearing aids and intensive speech therapy. CI is considered for infants who do not have significant cognitive delays and who demonstrate a substantial delay in language development, defined as being 5 months or more behind their age-matched peers. This method ensures that decisions regarding the timing of CI are made objectively. We have also analyzed CI outcomes, excluding children with CND, to isolate the effect of the age at surgery on the effectiveness of CI. Additionally, in South Korea, bilateral CI is covered by national insurance for infants under 12 months of age, provided they meet the hearing threshold criterion (a hearing loss worse than 90 dB) and exhibit an evident delay in language development. This eliminates SES as a factor in CI decisionmaking. Consequently, our study was able to assess the impact of CI timing without parental influence, offering a clear perspective on the advantages of early CI.
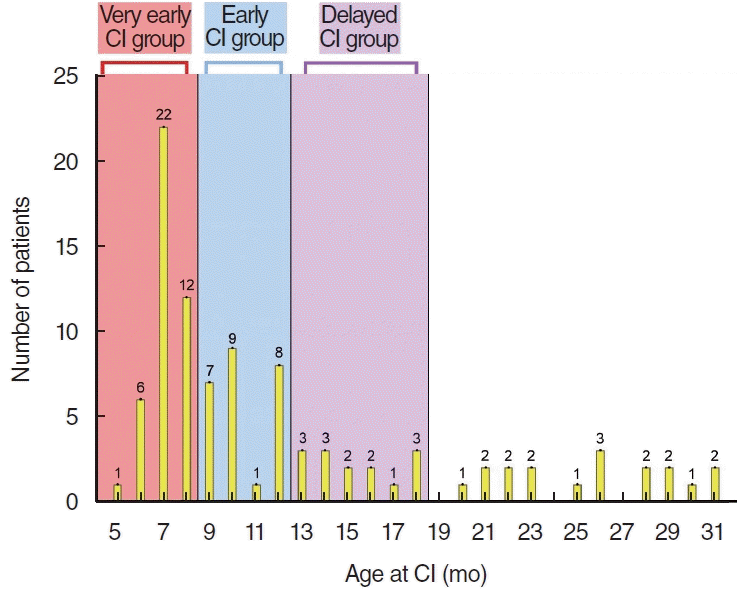
Based on the CI candidacy criteria at our center, we have observed a higher proportion of infants with profound deafness receiving CIs before the age of 9 months, as opposed to between 9 and 12 months. Specifically, a significantly larger number of infants with profound hearing loss underwent CI surgery before reaching 9 months of age (n=41) compared to those who underwent CI between 9 and 12 months (n=25). Consequently, comparing the surgical outcomes of these very early implantees with those who underwent CI between 9 and 12 months is of even greater significance.
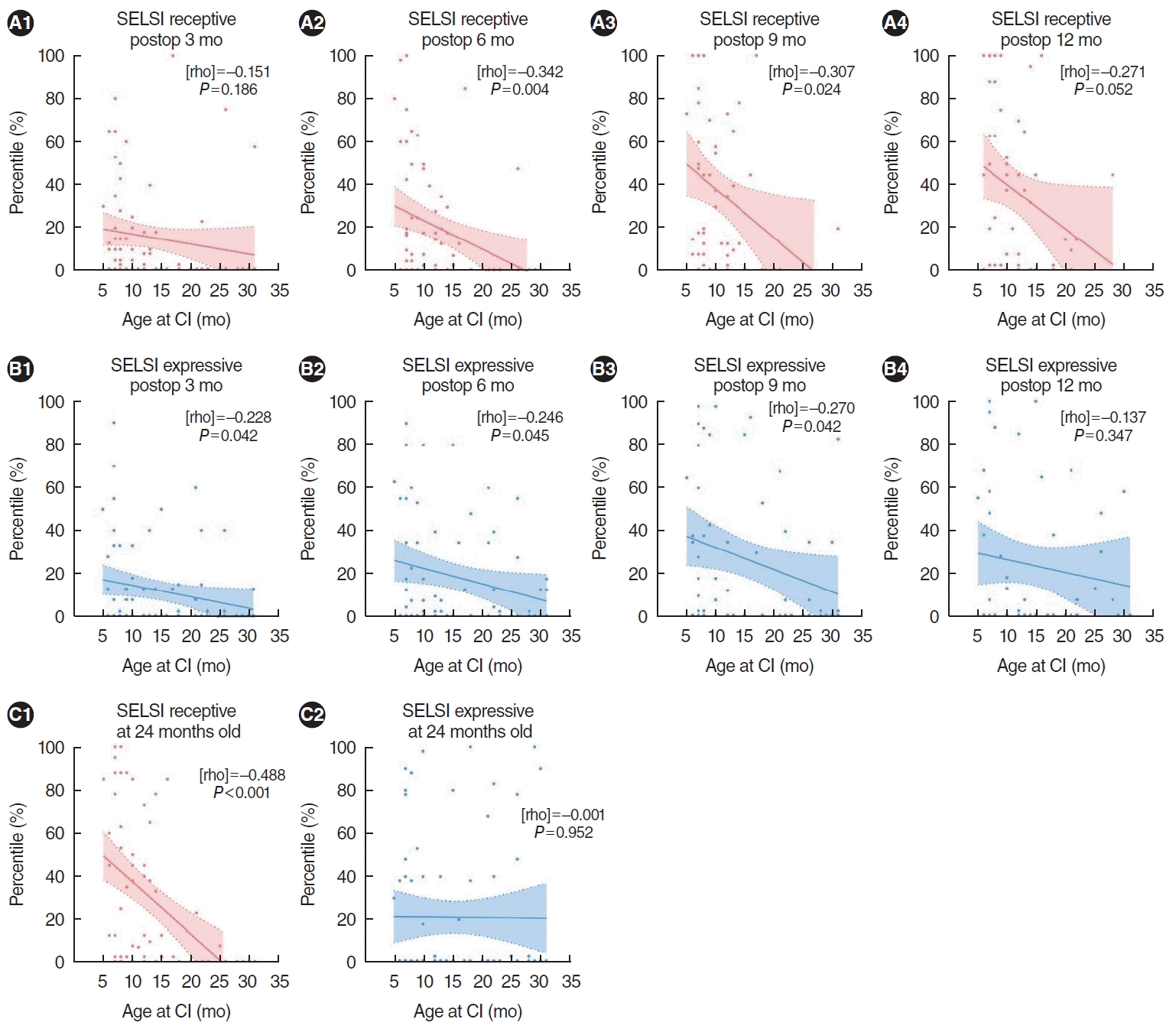
Our study warrants special attention as it provides an early trajectory of receptive and expressive speech development in CI recipients, particularly among those implanted very early (CI at <9 months of age) compared to early implantees (CI at 9–12 months of age) and those with delayed implantation (CI at 12–18 months of age). We discovered that infants who received implants before 9 months exhibited superior receptive language development compared to those implanted between 9 and 12 months, both after the first postoperative year and at two years of age. The very early group demonstrated the most rapid growth in receptive language starting from 3 months postoperation and matched the language development of their normal-hearing peers by 9 months post-operation. By age 2, they were the only group to achieve receptive language milestones comparable to those of normal-hearing children. Additionally, our analysis indicated that receiving a CI before 9 months is associated with better receptive language outcomes up to 1 year postoperation and at 2 years of age, while the benefits to expressive language were less pronounced. This underscores the benefits of implantation before 9 months for children with profound bilateral deafness and a known etiology. Correlation analysis between the SELSI receptive/expressive scores and the age at CI, ranging from as early as 5 months to at least 24 months, revealed consistent negative correlations at multiple postoperative benchmarks, particularly at 6 and 9 months post-surgery. Notably, at the age of 24 months, there was a significant inverse relationship between younger ages at CI and higher receptive language scores, a pattern not observed in the expressive scores. The data clearly underscore the critical importance of performing CI before the age of 9 months in children with profound bilateral deafness and a known favorable etiology. It specifically demonstrates that the advantages of CI performed at ages younger than 9 months persist up to at least the age of 2. Furthermore, very early CI has a more significant impact on the development of receptive language skills than expressive language skills during this period.
Consistent with our findings, Culbertson et al. [
10] demonstrated that implantees under 9 months of age rapidly improve in auditory skills postoperatively. By the age of 2 years, these children matched the auditory skills of their normal-hearing peers and continued to outperform those implanted later, at least until the age of 4 [
10]. Similarly, Karltorp et al. [
5] found that children implanted before 9 months exhibited language comprehension growth rates comparable to those of normal-hearing children, as measured by the Reynell scales, particularly up to the age of 4. It appears that initiating CI before 9 months positively impacts receptive language development (as per our study), auditory skills, and language comprehension, up until the ages of 2, 4, and 4 years, respectively. Since auditory skills precede spoken language development and predict speech production abilities [
33], it is reasonable to expect that expressive language improvements, benefiting from very early CI, may emerge later than receptive language gains and may not be fully evident by age 4. Of course, we cannot rule out the possibility that after the age of 4, children in both the early and delayed CI groups might catch up to the very early group, potentially leading to no significant differences in higher language skills in the long term. Indeed, studies such as that of Dunn et al. [
34] have analyzed long-term outcomes in children who received CIs before the age of 4 years, categorized by whether they were implanted before or after 2 years of age. They observed that the differences in long-term language outcomes between these groups diminished over time, suggesting that the influence of CI timing on language development decreases as children grow older [
34]. However, it remains uncertain whether this trend applies to CIs performed much earlier than 2 years of age. To clarify this, longer-term studies with larger cohorts, including a significant number of very early CI and early CI groups, are necessary in future research.
Indeed, recent studies have increasingly focused on the long-term impact of CI before 9 months of age on language development. Chweya et al. [
3] in the United States found that children who received CIs before 9 months of age exhibited superior receptive and expressive language development compared to those who were implanted between 9 and 12 months of age, after an average follow-up of 7.4 years. They observed a negative correlation between the age at implantation (ranging from 5 to 35 months) and language scores at the most recent follow-up, as evaluated by the Pre-school Language Scales Fourth and Fifth Editions (PLS-4 and PLS-5) and the Oral and Written Language Scales tests. This indicates that earlier CI (before 9 months) has a positive effect on long-term receptive and expressive language outcomes. Dettman et al. [
9] also reported better receptive language outcomes, as measured by the Categories of Linguistic Performance, in children who received their implants very early, at age 5. Karltorp et al. [
5] found that the correlation between age at CI and post-CI receptive and expressive vocabulary development did not become evident until the children were older, specifically at ages 6 and 8, respectively. Furthermore, the correlation with expressive language development appeared even later, at the age of 8, compared to the correlation with receptive language development. These findings are consistent with those from our study, which had a shorter follow-up period and did not yet show differences in expressive language development between the very early (before 9 months) and early (between 9 and 12 months) CI groups.
One intriguing point in this study is the comparison between the very early CI group and the delayed CI group. Examination of their pre-surgical hearing status revealed that the delayed CI group had significantly better preoperative speech development. Additionally, this group demonstrated a statistically significant better auditory threshold before CI, at least in one ear (
Table 2,
Supplementary Table 2). This finding helps explain why their CI was postponed. It is important to note that half of the individuals in the delayed CI group were diagnosed with enlarged vestibular aqueduct—a condition that is historically linked to a favorable prognosis after CI [
35]. However, longitudinal observations over the first postoperative year indicate that the very early CI group, though not statistically significant, tends to show higher receptive language development percentiles immediately following CI compared to the delayed CI group. By 24 months of age, the very early CI group reaches the median milestone for receptive language development, whereas the delayed CI group, despite better pre-surgery language and hearing status, does not. This pattern was not observed in the early CI group. This suggests that even when one ear has a hearing threshold between 80 and 90 dB (which is better than a profound degree), it may be wise to consider CI, at least unilaterally, before the age of 9 months to optimize receptive language outcomes. To incorporate this approach into clinical practice in South Korea, changes to the government’s pediatric CI national insurance criteria are required. Currently, the criteria stipulate a hearing threshold greater than 90 dB bilaterally for children under 12 months of age.
The strengths of this study include the establishment of clear, objective criteria for CI timing in infants, with decisions made solely based on these criteria. This approach minimizes the influence of parental SES and willingness on rehabilitation outcomes. All surgeries were performed by the same surgeon at a single center, and the same type of electrodes was used in most cases, which reduces bias and variables other than the age at CI. Cases with CND, which are known for less favorable outcomes, were excluded from the performance analysis. However, one limitation of this study is that it only compares outcomes up to the age of 2 years. This necessitates long-term follow-up to determine whether the superior effects of very early CI (under 9 months) over early CI (9 to 12 months) are sustained over time. Additionally, we compared the language development scores of all children in groups A, B, and C. Due to the small number of subjects, particularly in group C, we were unable to perform a comparison of language development scores among subjects who underwent simultaneous CI. While this may be considered a potential limitation of the study, the majority of subjects in all groups received simultaneous implants. Therefore, we do not anticipate a significant difference in language development outcomes when comparing only those who underwent simultaneous CI. For those who did not receive simultaneous implants but underwent sequential or unilateral CI, we believe that their inclusion in the comparison did not bias the results in favor of group A. This is because individuals, especially in groups B or C, likely had more residual hearing or more advanced language development prior to receiving their CI.
In conclusion, we established criteria for determining the appropriate timing of CI in infants, which are based on objective assessments of hearing and language development status. These criteria are independent of the parents’ financial capacity or willingness to pursue rehabilitation, thanks to the provision of government insurance coverage. According to our findings, which are the first of their kind to our knowledge, infants who receive off-label, very early CI (under 9 months of age) demonstrate more advanced receptive and expressive language development in the first year post-implantation than those who receive early CI (between 9 and 12 months of age). Although the gains in expressive language are less pronounced during this period, they are still noteworthy. Remarkably, only the very early CI group (under 9 months of age) shows significant improvement in receptive language beginning at 3 months post-CI. These infants rapidly reach the level of their normal-hearing peers by 9 months postoperatively and maintain this advanced level at least until the age of two. It is also significant that the very early CI group tends to exhibit superior receptive language development during this period compared to the delayed CI group, where implantation is postponed until after 12 months of age due to the presence of significant residual hearing and advanced preoperative language development. These findings support the consideration of earlier CI in infants with documented etiologies and speech delays, without being restricted to the pediatric FDA labeling criteria, which currently recommend implantation at 9 months of age or older.