 |
 |
- Search
AbstractObjectives.Patients with obstructive sleep apnea syndrome (OSAS) have impaired responses to inspiratory resistive loading during sleep. This may be due, in part, to a change in the upper airway sensation. Therefore, we hypothesized that patients with OSAS have diminished upper airway sensation due to snoring.
Methods.A total of 53 participants were selected based on clinical evaluation and polysomnography. Two-point discrimination was measured with modified calipers in the tongue and soft palate.
Results.A total of 10 participants were included in the control group, 12 participants in the simple snoring group, and 27 participants in the OSAS group. There were 12 patients in the impaired sensation group of the OSAS group. On comparing polysomnography, patients with impairment of their palatal sensory input in two-point discrimination (TPD) had a more protracted duration of the longest snoring episode than those with simple snoring and normal sensation. Patients with decreased sensory input in TPD had longer average duration of snoring episodes and relative snoring time than those with simple snoring and normal sensory input in cold uvular TPD. Comparison of the cold uvular TPD for normal sensation and impaired sensation in patients with OSAS after treatment showed a different trend.
Conclusion.Impaired sensation of the soft palate was correlated with the longest snoring episode duration, average snoring episode duration, and relative snoring time. It is helpful in detecting the early stage of neural degradation in OSAS patients by assessing snoring components of polysomnography and TPD in the soft palate.
Apneic episode in obstructive sleep apnea (OSA) starts with the collapse of the upper airway during sleep, and resolves when stimuli generated during apneas cause a brief arousal and reopening of the airway. In general, obstructive sleep apnea syndrome (OSAS) is progressive over time and its prevalence increases with age [1,2]. Svanborg [3] proposed and pursued the hypothesis on the pathogenesis of OSA that long-standing snoring-induced vibrations cause neurogenic lesions in upper airway tissues, and then cause damage to the reflex circuits which keep the upper airway open during inspiration [3]. This could be critical during sleep because the muscle tone is normally reduced. Several studies have supported this hypothesis by confirming upper airway neuropathy in OSAS patients [3-5]. Other research groups have also supported the hypothesis with contributing data [6-8]. In these studies, various methods for measuring local sensory neuropathy have been used, such as vibration [6], 2-point discrimination [7], and air-pressure pulses [8]. However, no attempts have been made to correlate the degree of sensory deficit in the soft palate with the duration of snoring.
We have demonstrated measurement of warm and cold two-point detection (TPD) with the noninvasive method of levels (MLE). In the MLE, stimuli of predetermined intensities are used and the test subject responds after the stimulus has been given to confirm whether it was detected or not. The purpose of this study was to use this method to compare sensory deficit in the soft palate among untreated subjects with OSAS, and to correlate these data with the apnea-hypopnea index (AHI) and the duration of snoring.
Patients with sleep disorders and normal subjects who fulfilled the following clinical and polysomnographic inclusion criteria were included: (1) clinical criteria, age between 17 years and 65 years (or premenopausal women); absence of neurologic, cardiovascular, pulmonary, or other chronic illness; no prior surgery of the nose and palate; no current drug intake; modified Friedman stage I; oropharyngeal obstruction during the Müller maneuver; and no prior treatment of sleep-disordered breathing; (2) polysomnographic recording criteria, patients with OSAS should have an AHI ≥5/hr of sleep; however, simple snorers should have an AHI <5/hr of sleep with concomitant snoring (relative snoring time ≥5%) on polysomnography; otherwise, the control group should have an AHI <5/hr of sleep without a snoring event (relative snoring time <5%) during polysomnography; and (3) subjects should provide informed consent. Exclusion criteria were any previous treatment for snoring or OSA, previous upper airway surgery excluding remote tonsillectomy, previous cerebrovascular accident, diabetes, any neuropathy or active neurologic disease, and recent upper respiratory tract infection, excessive gag reflex, or a very small oropharyngeal cavity at the time of proposed testing, which precluded sensory testing. All OSA patients underwent lateral pharyngoplasty and tonsillectomy with sparing of the uvula. We evaluated the initial TPD on the same day the patient underwent polysomnography before surgery. Also, the TPD during follow-up was measured after 3 months of surgery. Approval for the trial was granted by Konkuk University Hospital Ethics Committee (KUH1110017).
All subjects with an initial evaluation who met the inclusion criteria underwent nocturnal polysomnography. The following variables were monitored: electroencephalogram (EEG), electrooculogram, chin electromyogram, leg electromyogram, heart rate (modified V2 lead), and body position. Respiration was evaluated with nasal cannula pressure transducer system, polygraphic recording with esophageal pressure (PES) transducer, thoracic bands, abdominal bands, pulse oximeter, and neck microphone. Recordings were scored following the international criteria for sleep/wake with identification of short (≥3 seconds) arousals; tabulation of apneas, hypopneas; and obstructive, mixed events, and central events following the criteria of the American Academy of Sleep Medicine [9]. There was also scoring of short visual arousals (≥3 seconds) secondary to ‘abnormal breathing efforts.’ This scoring was performed using PES recording with recognition of ‘PES crescendos’ and ‘sustained abnormal respiratory effort’ as previously described [10], in addition to the nasal cannula/pressure transducer system with recognition of respiratory events of <10 seconds duration but terminated by a visual EEG arousal of ≥3 seconds (arousal with abnormal breathing effort). Snoring components consisted of relative snoring time (%), average snoring episode duration (minutes), longest snoring episode (minutes), total snoring time (minutes), and number of snoring episodes. These components were calculated on polysomnography (Somnologica Studio, Embla Systems Inc., Broomfield, CO, USA).
A special compass-like device was built by one of the authors (GTK) (Fig. 1). When the two branches were in close contact, the distance was 0.2 mm in diameter. A screw system allowed opening of the two branches to keep the two branches fixed at the selected distance. A thermode was applied to the soft palate and the anterior tongue. TPD was determined with the MLE [11]. The apparatus was set at a starting temperature of 43°C and 0°C for soft palate testing and anterior tongue testing, respectively. Then, each subject was asked to indicate 1 or 2 points by holding up the appropriate number of fingers to confirm whether or not he/she perceived a sensation of cold (warm) as soon as they perceived a sensation of cold (warm) [12]. The first stimulus step size was 6 mm. If the subject did not perceive the first stimulus, the next stimulus was increased in the 3-mm size until the subject responded (first stimulus 6 mm, second stimulus 9 mm, third stimulus 12 mm etc.). If the subject first perceived a stimuli sensation at for example 6 mm, new stimuli, with subtraction of 2-mm step size from the previous round, were repeated until a ‘no’ response was obtained (6, 4, 2, 1 mm etc.). If the subject then perceived stimuli at for example 1.0 mm, the intensity of the next stimulus was increased again in steps half the size of those in the previous round (1, 2, 3 mm etc.) until a ‘yes’ response was obtained. The time intervals between the four stimuli were randomized to last between 4 and 6 seconds. The length of each test was measured by one of the authors.
As there was no available definition of ‘impaired sensation of the soft palate,’ we defined it as the length of discrimination distance of the soft palate is longer than one-half of the length of discrimination distance of the tongue individually.
Descriptive statistics were used for population and group characteristics. Correlations between soft palate TPD and age, AHI, and snoring components of polysomnography were evaluated with group analysis performed using Kruskal-Wallis test. Receiver operating characteristic (ROC) curve was applied for the cutoff value of sensory change [13]. To compare between the pre-treatment TPD and post-treatment TPD, the Wilcoxon signed rank test was performed. All data were analyzed with IBM SPSS statistical software ver. 20.0 (IBM Co., Armonk, NY, USA). Statistical significance was set at P<0.05.
A total of 53 individuals were included in the study. Two subjects (two men) could not participate in TPD testing due to strong gag reflexes, and two subjects (both men) had technically unsatisfactory recordings. Characteristics of the remaining forty-two men and seven women included in the analysis are summarized in Table 1. The age and body mass index (BMI) were significantly different in the control group, but no difference was observed between the simple snoring group and the OSAS group, and AHI in the OSAS group was higher than that in the other groups. Compared with the control group, the mean TPD distance at 0°C and 43°C was longer in the OSAS group, but it was not significantly different. There were no significant differences in age and TPD between the OSAS group and the control group.
Some subjects in the OSAS group displayed longer TPD distance than the other subjects in the same group. Furthermore, their snoring components tended to increase. We investigated the cutoff value for defining the sensory change. The mean length of 2.5 mm in the soft palate cold TPD test obtained by the ROC curve analysis was the cutoff value with 91.7% sensitivity and 85.7% specificity (area under curve=0.901, P<0.001) (Table 2).
There were 15 patients in the normal sensation group and 12 patients in the impaired sensation group (Fig. 2). There were no significant differences in age, BMI, and AHI between the two groups (Table 3). The results of the 43°C TPD test in the anterior tongue and soft palate showed that the simple snoring group had a significantly shorter distance than the other groups (P=0.004, P<0.001, respectively). Also, the results of the soft palate cold TPD test demonstrated that the impaired sensation group had a significantly longer distance than the other groups (P<0.001).
On polysomnography, the mean AHI in the OSAS group was higher, and varying results were observed for snoring components. Average snoring episode duration, longest snoring episode, and relative snoring time in the impaired sensation group were significantly longer than those in the normal sensation group (P=0.043, P=0.010, and P=0.032, respectively) (Figs. 3–5) and lowest O2 saturation was significantly different in the simple snoring group (P=0.009).
Compared to the mean value before treatment, the mean value after treatment did not significantly change for cold TPD in the normal sensation group (P=0.291) (Fig. 5). In the normal sensation group, the mean±SD before treatment was 2.1±1.3, and the mean±SD after treatment was 2.3±1.2. In the impaired sensation group, the mean±SD before treatment was 4.6±1.6, and the mean±SD after treatment changed to 5.3±2.6 without statistical significance (P=0.392). But, the difference between the normal and impaired sensation groups in cold TPD after treatment was statistically significant (P=0.001).
Segmental demyelination and axonal degeneration of afferent neurons, such as α-fibers, lead to sensory impairment. These lesions are responsible for slowing of impulse conduction [14]. Friberg et al. [15,16] provided evidence for local neurogenic lesions in heavy snorers and OSAS patients [3]. Other studies supported the hypothesis that snoring is associated with the histologic alterations; Kimoff et al. [6] suggested abnormal responses in snorers. Our data partially agree with these studies. The patients in the simple snoring group did not show any sensory change in the soft palate compared to those in the control group, but the patients in the OSAS group showed sensory change in the soft palate.
On comparing TPD in cold and warm temperatures, there was no significant difference between the OSAS group and the control group. This finding is similar to that in the report by Guilleminault et al. [7], although we did not separate the upper airway resistance syndrome subjects and the OSAS subjects. The previous authors explained that there was not enough snoring during the night and/or the number of years spent snoring was insufficient in patients with OSAS to induce the neurologic lesions. In contrast to the present study, Sunnergren et al. [17] reported a significant positive correlation between both estimated periods of snoring and they objectively evaluated the degree of sleep-disordered breathing with the degree of impaired sensation of the soft palate at an individual level. However, in this prior study, snoring was not recorded objectively.
Our study strengthens the hypothesis that long-standing, snoring-induced vibrations could cause impaired sensation of the soft palate, and that sensory change in the soft palate might be involved in the pathologic progression often seen in OSA. It is well-known that long-term exposure to low-frequency vibration causes peripheral nerve injury in humans [18]. In our study, we determined 2.5 mm in cold TPD as the cutoff value for impaired sensation of the soft palate, which could possibly be used for early detection of palatal peripheral neuropathy in OSAS subjects. Previous studies only made a comparison among non-snorers, snorers, and OSA subjects [6,7,17] without the use of a cutoff value. In our study, there was no difference between preoperative TPD and postoperative TPD. Therefore, further studies with long-term observation and larger sample size are needed to verify our suggestion because a short follow-up period and a small sample size were used in this study.
Long-standing vibrations not only cause sensory neuropathy due to thermal stimuli but also due to mechanical stimuli [19,20]. In agreement with this phenomenon, an interesting finding of the present study was that the OSAS patients with greater longest snoring time, average snoring episode duration, and relative snoring time on polysomnography tended to have impaired sensation of the soft palate. This finding was also consistent with previous studies [6,7,15-17]. In spite of the conception that the subocclusive stage in snoring precedes the development of OSA [21], there was a broad spectrum ranging from mild to severe snorers among OSAS subjects. This indicated that it was not always a one-way progression from snoring to OSA. We speculate that vibration-induced snoring may have played a more important role in sensory change in the soft palate than oxygen desaturation in OSAS subjects. The clinical importance of sensory change in the soft palate was not totally deciphered in the present study, but based on the concept that the continuous positive airway pressure (CPAP) treatment created airflow buttress that reduced snoring, CPAP would be helpful in preventing impaired sensation of the soft palate.
The limitations of this study were its small sample size and the lack of comparison of clinical symptoms. Long-term, follow-up observations are needed. The limitation of the present study also included unknown duration of OSAS because subjects could not report the accurate time of onset of symptoms. A further study assessing the change in snoring components after treatment is needed. Another limitation that should be considered is that psychophysical methods, such as two-point discrimination testing, require active participation of the subjects. Therefore, the results could have been affected by psychological confounding factors. Also, a comparison of the tools used in this study and the previous study was not performed. Although the devices used in the previous study had a relatively same scale range as our device, a further study comparing the published tools is needed to determine the accuracy of TPD.
In conclusion, impaired sensation of the soft palate is correlated with longest snoring episode duration, average snoring episode duration, and relative snoring time, and hence, it could be useful in detecting the early stage of neural degradation in OSAS patients through assessment of snoring components of polysomnography and two-point discrimination in the soft palate.
▪ Patients with impaired palatal sensory input in two-point detection had more protected duration of the longest snoring episode.
▪ Those also had longer average duration of snoring episodes and relative snoring time.
▪ Impaired palatal sensation was well correlated with all the snoring measurements.
ACKNOWLEDGMENTSThis work was supported by the National Research Foundation of Korea (NRF) grant funded by the Korea government (Ministry of Science, ICT and Future Planning) (No. NRF-2013R1A1A2009589).
Fig. 1.Modifi ed caliper device. Special compass-like device consisting of temperature display and control buttons (A), scale (B), and thermode with a screw system (arrow). 
Fig. 2.Demographics of subjects. A total of 49 patients were enrolled. The number of subjects in the control group was 10, that in the simple snoring group was 12, that in the normal sensation group of the OSAS group was 15, and that in the impaired sensation group of the OSAS group was 12. AHI, apnea-hypopnea index; TPD, two-point detection; OSAS, obstructive sleep apnea syndrome. 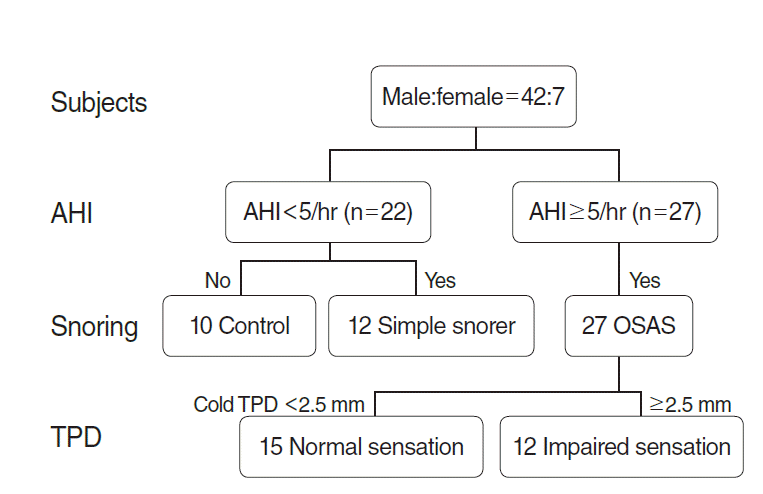
Fig. 3.(A) Longest snoring episode (min). The impaired sensation group of the OSAS group showed a significant difference. The mean±SD was 15.6±10.3 in the impaired sensation group, 7.4±5.6 in the simple snoring group, and 3.9±4.5 in the normal sensation group of the OSAS group. Symbol (*) indicates statistical significance. (B) Average snoring episode duration (minute). The impaired sensation group of the OSAS group showed a significantly longer duration than the other groups. The mean±SD was 0.7±0.4 in the impaired sensation group, 0.4±0.2 in the simple snoring group, and 0.4±0.3 in the normal sensation group of the OSAS group. Symbol (*) indicates statistical significance. OSAS, obstructive sleep apnea syndrome. 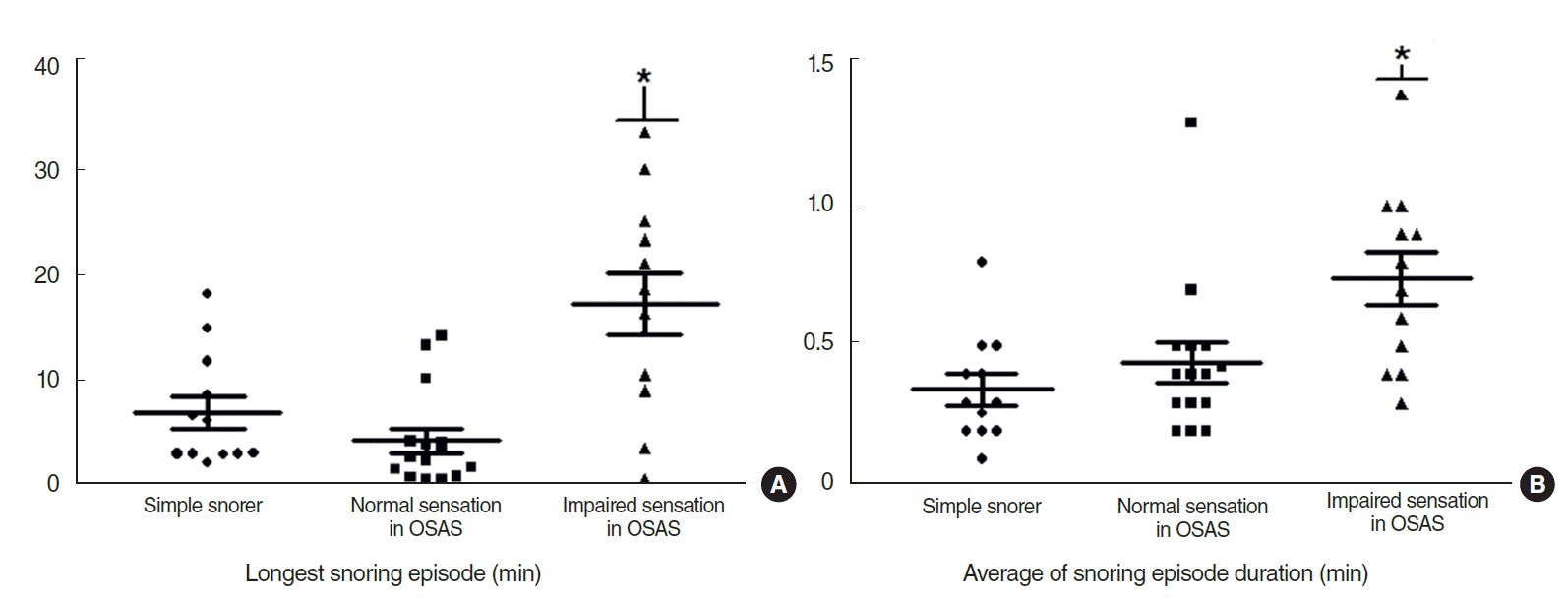
Fig. 4.(A) Relative snoring time (%). The mean±SD was 16.8±4.8 in the simple snoring group, 18.2±11.3 in the normal sensation group, and 28.7±10.8 in the impaired sensation group of the OSAS group. The impaired sensation group showed a significant difference. Symbol (*) indicates statistical significance. (B) Lowest O2 saturation (%). The mean±SD was 86.9±3.5 in the simple snoring group, 80.7±6.8 in the normal sensation group, and 79.9±6.5 in the impaired sensation group of the OSAS group. There was no significant difference between the normal and impaired sensation groups except in the simple snoring group. Symbol (*) indicates statistical significance. OSAS, obstructive sleep apnea syndrome. 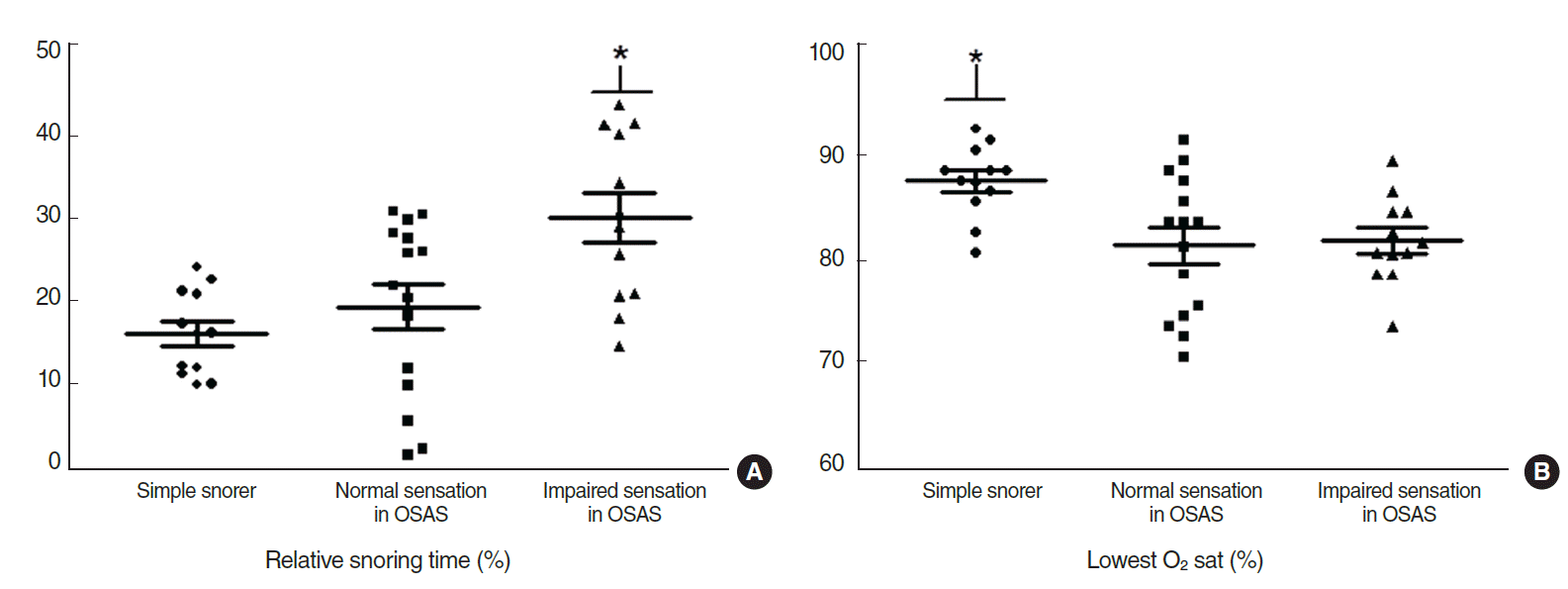
Fig. 5.Alteration of cold TPD between the normal and impaired sensation groups of the OSAS group. Compared to the mean before treatment, the mean after treatment in the normal sensation group did not change signifi cantly for cold TPD (P=0.291). In the impaired sensation group, the cold uvular TPD before treatment was increased without statistical signifi cance (P=0.392). Symbol (*) indicates statistical signifi cance. NS_before, the normal sensation group before treatment; NS_after, the normal sensation group after treatment; IS_before, the impaired sensation group before treatment; IS_after, the impaired sensation group after treatment; TPD, two-point detection; OSAS, obstructive sleep apnea syndrome. 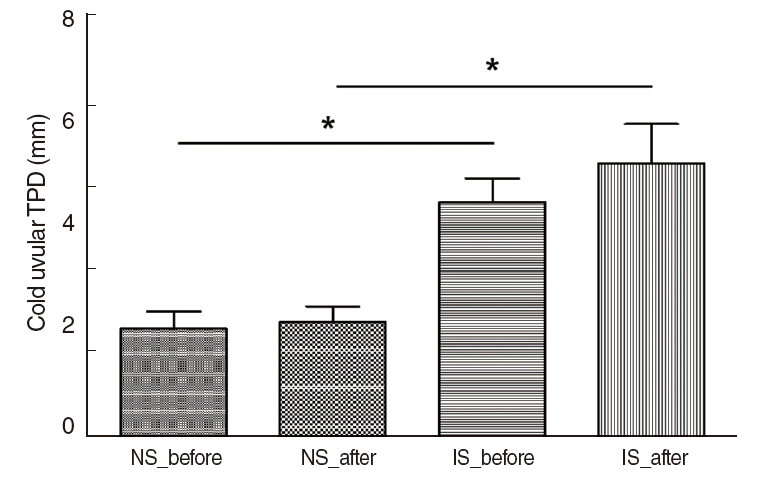
Table 1.Subject characteristics
Table 2.Sensitivity and specificity of cutoff point for diagnosing impaired sensation group at different thresholds
Table 3.Snoring components between the normal sensation group and the impaired sensation group
REFERENCES1. Silva GE, An MW, Goodwin JL, Shahar E, Redline S, Resnick H, et al. Longitudinal evaluation of sleep-disordered breathing and sleep symptoms with change in quality of life: the Sleep Heart Health Study (SHHS). Sleep. 2009 Aug;32(8):1049-57.
2. Berger G, Berger R, Oksenberg A. Progression of snoring and obstructive sleep apnoea: the role of increasing weight and time. Eur Respir J. 2009 Feb;33(2):338-45.
3. Svanborg E. Impact of obstructive apnea syndrome on upper airway respiratory muscles. Respir Physiol Neurobiol. 2005 Jul;147(2-3):263-72.
4. Friberg D, Ansved T, Borg K, Carlsson-Nordlander B, Larsson H, Svanborg E. Histological indications of a progressive snorers disease in an upper airway muscle. Am J Respir Crit Care Med. 1998 Feb;157(2):586-93.
5. Hagander L, Harlid R, Svanborg E. Quantitative sensory testing in the oropharynx: a means of showing nervous lesions in patients with obstructive sleep apnea and snoring. Chest. 2009 Aug;136(2):481-9.
6. Kimoff RJ, Sforza E, Champagne V, Ofiara L, Gendron D. Upper airway sensation in snoring and obstructive sleep apnea. Am J Respir Crit Care Med. 2001 Jul;164(2):250-5.
7. Guilleminault C, Li K, Chen NH, Poyares D. Two-point palatal discrimination in patients with upper airway resistance syndrome, obstructive sleep apnea syndrome, and normal control subjects. Chest. 2002 Sep;122(3):866-70.
8. Nguyen AT, Jobin V, Payne R, Beauregard J, Naor N, Kimoff RJ. Laryngeal and velopharyngeal sensory impairment in obstructive sleep apnea. Sleep. 2005 May;28(5):585-93.
9. Choi JH, Jun YJ, Kim TH, Lee HM, Lee SH, Kwon SY, Choi H, et al. Effect of isolated uvulopalatopharyngoplasty on subjective obstructive sleep apnea symptoms. Clin Exp Otorhinolaryngol. 2013 Sep;6(3):161-5.
10. Guilleminault C, Poyares D, Palombini L, Koester U, Pelin Z, Black J. Variability of respiratory effort in relation to sleep stages in normal controls and upper airway resistance syndrome patients. Sleep Med. 2001 Sep;2(5):397-405.
11. Shy ME, Frohman EM, So YT, Arezzo JC, Cornblath DR, Giuliani MJ, et al. Quantitative sensory testing: report of the Therapeutics and Technology Assessment Subcommittee of the American Academy of Neurology. Neurology. 2003 Mar;60(6):898-904.
12. Tapia IE, Bandla P, Traylor J, Karamessinis L, Huang J, Marcus CL. Upper airway sensory function in children with obstructive sleep apnea syndrome. Sleep. 2010 Jul;33(7):968-72.
13. Akobeng AK. Understanding diagnostic tests 3: receiver operating characteristic curves. Acta Paediatr. 2007 May;96(5):644-7.
14. Mackenzie RA, Skuse NF, Lethlean AK. A micro-electrode study of peripheral neuropathy in man. Part 1. Responses to single graded stimuli. J Neurol Sci. 1977 Nov;34(2):157-74.
15. Friberg D, Gazelius B, Hokfelt T, Nordlander B. Abnormal afferent nerve endings in the soft palatal mucosa of sleep apnoics and habitual snorers. Regul Pept. 1997 Jul;71(1):29-36.
16. Friberg D, Gazelius B, Lindblad LE, Nordlander B. Habitual snorers and sleep apnoics have abnormal vascular reactions of the soft palatal mucosa on afferent nerve stimulation. Laryngoscope. 1998 Mar;108(3):431-6.
17. Sunnergren O, Brostrom A, Svanborg E. Soft palate sensory neuropathy in the pathogenesis of obstructive sleep apnea. Laryngoscope. 2011 Feb;121(2):451-6.
18. Takeuchi T, Futatsuka M, Imanishi H, Yamada S. Pathological changes observed in the finger biopsy of patients with vibration-induced white finger. Scand J Work Environ Health. 1986 Aug;12(4 Spec No):280-3.
19. Stromberg T, Dahlin LB, Lundborg G. Hand problems in 100 vibration-exposed symptomatic male workers. J Hand Surg Br. 1996 Jun;21(3):315-9.
|
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||







